AtaGenix Laboratories
AtaGenix Laboratories
Release time: 2026-01-14 View volume: 667
Project Snapshot — An Advanced Science study (IF: 14.3) identified the PAD1–AKT2(R202)–CEBPβ axis as a driver of ovarian cancer stem-like cell (OCSLC) stemness and cisplatin resistance. PAD1 citrullinates AKT2 at Arg202, maintaining phosphorylation site exposure and activating PI3K-AKT–CEBPβ signaling. AtaGenix provided a custom site-specific Cit202 antibody that enabled direct detection of AKT2 R202 citrullination in co-IP, WB, and IHC workflows.
Based on: Xue T et al., Advanced Science, 2025;12(39):e01014 — DOI: 10.1002/advs.202501014
Ovarian cancer remains one of the deadliest gynecologic malignancies, with 5-year survival dropping to ~32% at distant stage. Recurrence and chemotherapy resistance are major clinical challenges, with ovarian cancer stem-like cells (OCSLCs) as key resistance drivers. The regulatory role of peptidylarginine deiminase 1 (PAD1) in OCSLCs had not been defined, highlighting the need for mechanistic targets to overcome therapeutic bottlenecks.
Using RNA-seq, co-immunoprecipitation, xenograft models, and clinical validation (33 ovarian cancer vs. 33 ovarian cyst tissues), the authors identified the PAD1–AKT2/R202 citrullination–CEBPβ axis as a regulatory mechanism supporting OCSLC stemness. PAD1-axis activation was enriched in cisplatin-resistant OVCAR3-CisR cells, and pharmacological PAD1 inhibition re-sensitized resistant cells to cisplatin and reduced tumor burden in vivo.
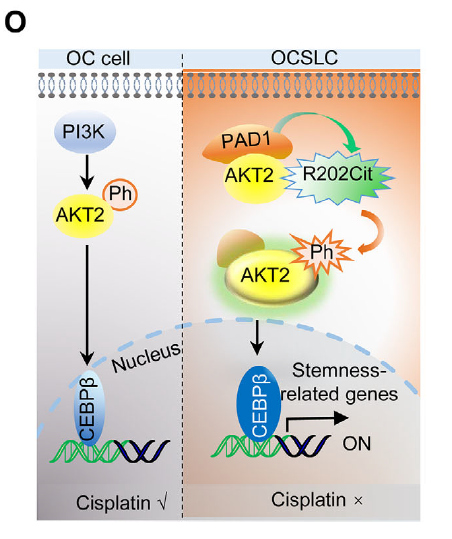
Fig. 1. Mechanistic model: PAD1 citrullinates AKT2 at R202, activating PI3K-AKT–CEBPβ signaling to maintain OCSLC stemness and cisplatin resistance. Adapted from Xue et al., Adv Sci 2025.
PAD1 Upregulation: PAD1 is highly expressed in OCSLCs (CD133+ sorted cells) and specifically binds to the AKT2 kinase domain (152–481 aa).
Citrullination Event: PAD1 catalyzes AKT2 citrullination at Arg202, maintaining exposure of phosphorylation sites (S474/T309) and promoting kinase activity.
Downstream Activation: Enhanced AKT2 activity upregulates transcription factor CEBPβ, which activates stemness genes (CD133, SOX2) to sustain self-renewal and tumor-initiating capacity.
Therapeutic Vulnerability: Pharmacological PAD1 inhibition combined with cisplatin re-sensitizes resistant cells and reduces tumor burden in xenograft models.
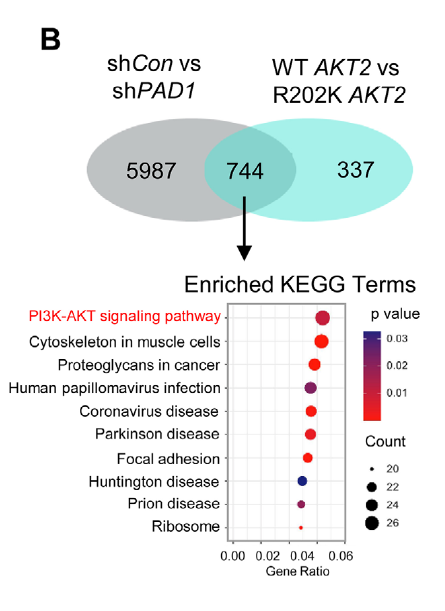
Fig. 2. Validation of PAD1–AKT2 interaction by co-immunoprecipitation. Adapted from Xue et al., Adv Sci 2025.
AtaGenix developed a custom site-specific antibody recognizing AKT2 Arg202 citrullination (Cit202), designed to minimize cross-reactivity with AKT homologs or unmodified AKT2. The Cit202 antibody enabled clear readouts in co-immunoprecipitation and immunohistochemistry workflows, providing direct evidence that PAD1 regulates AKT2 R202 citrullination and facilitating downstream exploration of modification-linked phenotypes and biomarker potential.
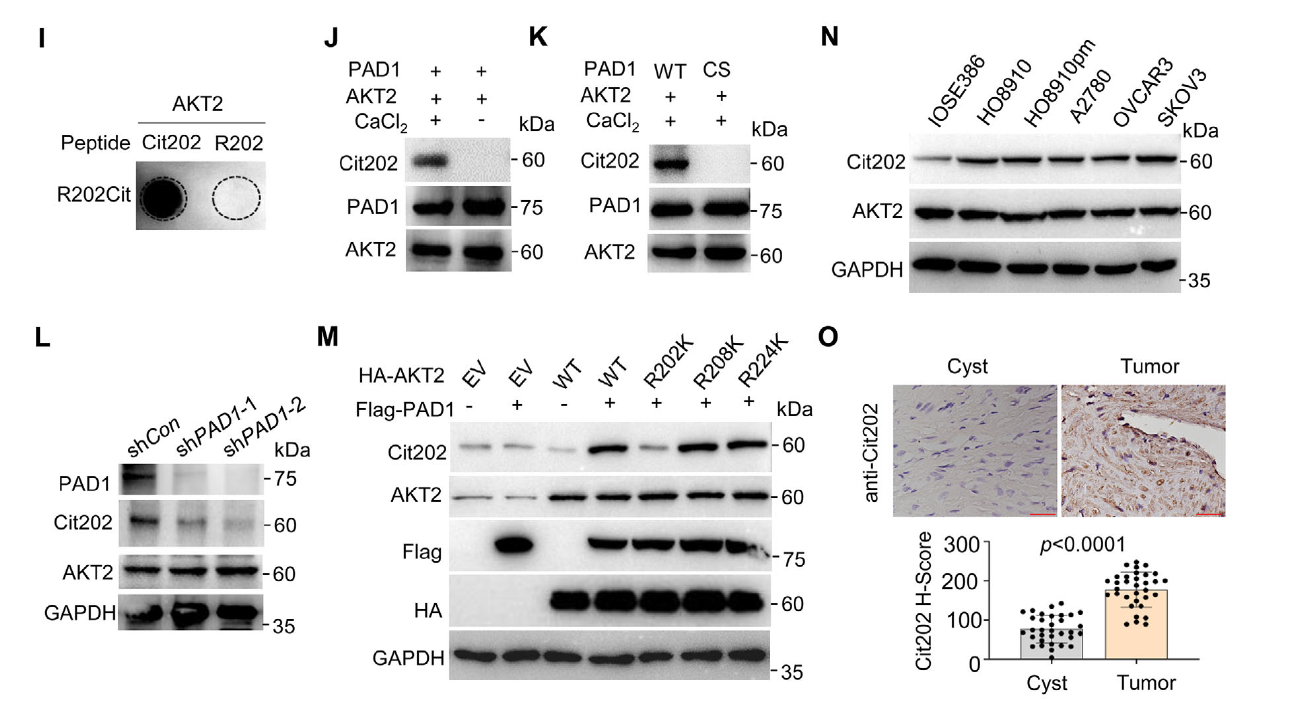
Fig. 3. AtaGenix Cit202 antibody detection results: site-specific recognition of AKT2 R202 citrullination in OCSLC contexts. Adapted from Xue et al., Adv Sci 2025.
14.3
Impact Factor (Adv Sci)
Cit202
Custom Site-Specific Ab
co-IP + IHC
Validated Applications
66
Clinical Samples Validated
Why This Matters
This study identifies citrullination — a relatively understudied post-translational modification — as a functional regulator of cancer stemness via the PAD1–AKT2(R202)–CEBPβ axis. The AtaGenix Cit202 antibody was the essential detection tool: no commercial antibody exists for site-specific AKT2 R202 citrullination, making this a de novo custom development that directly enabled the study's mechanistic conclusions. The finding that PAD1 inhibition re-sensitizes cisplatin-resistant ovarian cancer cells opens a stemness-oriented intervention strategy with translational potential.
Xue T, Liu X, Song C, et al. Citrullination of AKT2 catalyzed by PAD1 facilitates the maintenance of stemness characteristics of ovarian cancer stem-like cells. Advanced Science. 2025;12(39):e01014. DOI: 10.1002/advs.202501014
For research use only. This page provides scientific literature interpretation and does not constitute medical advice or treatment recommendations.
Need custom site-specific antibodies for novel post-translational modifications (citrullination, lactylation, methylation)? AtaGenix delivers modification-specific antibody programs with rigorous epitope design and multi-application validation.
Talk to Technical SupportResponse within 24 hours
Contact Us
+86-27-6552-3339
info@atagenix.com
Building C, R & D Building, No. 666, Shendun 4th Road, Donghu New Technology Development Zone, Wuhan

