AtaGenix Laboratories
AtaGenix Laboratories
AtaGenix's Rapid Hybridoma platform delivers validated monoclonal antibodies in as few as 59 days from immunization to frozen hybridoma clones. Our proprietary electrofusion technology ensures high fusion efficiency and rapid screening, giving you stable, renewable antibody sources for research, diagnostics, and therapeutic development.
With over 15 years of hybridoma experience and a >90% project success rate (per pre-agreed acceptance criteria), AtaGenix is trusted by global pharma, biotech, and academic partners for high-quality custom monoclonal antibody generation.
Our milestone-based project governance ensures full transparency: you approve at each checkpoint before moving forward. All deliverables include frozen hybridoma cells (≥5 vials), purified antibody (1–2 mg), ELISA validation data, isotype report, and Certificate of Analysis (COA).
High Fusion Efficiency
Electrofusion platform with superior fusion rates — more viable clones for screening
59-Day Rapid Delivery
Immunization to validated hybridoma clones in 59 days with milestone checkpoints
Complete Deliverables
Frozen cells, purified antibody (1–2 mg), ELISA data, isotype report, COA, and full IP
Whether you need a complete antibody development campaign or sequencing of existing hybridoma lines, AtaGenix delivers with precision and speed.
Standard Hybridoma Package
Timeline: 59 days | Mouse & Rat Hosts
Complete turnkey service from antigen to validated hybridoma clones:
| ✓ Customized immunization protocol (mouse/rat) | ✓ Electrofusion & ELISA screening |
| ✓ Subcloning to monoclonality | ✓ Isotype determination |
| ✓ Frozen cell stocks (≥5 vials) | ✓ Purified antibody (1–2 mg) + COA |
Premium Hybridoma Package
Everything in Standard, plus advanced characterization
| ✓ Full VH/VL sequencing | ✓ Western blot & IHC validation |
| ✓ Epitope mapping (optional) | ✓ Ascites or recombinant production |
| ✓ Extended cell banking | ✓ SPR/BLI affinity measurement (optional) |
Hybridoma Sequencing & Recombinant Expression
Already have hybridoma lines? We offer high-accuracy variable region and full-length sequencing with recombinant expression using our proprietary pATX3.0 vector.
| ✓ Variable region + full-length sequencing | ✓ CDR region analysis report |
| ✓ Recombinant expression: 200–500 mg/L yield | ✓ Turnaround: 2 weeks |
| ✓ Accepts cultured cells or frozen cell lines | |
| Your Need | Why Hybridoma |
| Long-term renewable supply | Frozen cell stocks provide indefinite antibody production with consistent lot-to-lot performance |
| Matched antibody pairs | Large screening panels naturally identify capture/detection pairs for sandwich ELISA and lateral flow |
| Diagnostic development | Full-length IgG with native Fc function, ideal for immunoassay and IVD applications |
| Budget-sensitive projects | Most cost-effective route to validated monoclonal antibodies for standard immunogenic targets |
59
Days to Delivery
>95%
Purity (SEC-HPLC)
10-7–10-9
Affinity KD (M)
100%
Full IP Ownership
Timeline counted from sample receipt. Actual duration depends on antigen availability and immune response. Milestone-based delivery with pre-agreed acceptance criteria. Quote-based pricing.
Our streamlined 59-day process moves through four clearly defined milestones with client sign-off at each gate.
01
Immunization
Week 1–4
Customized protocol
Mouse or rat hosts
Flexible antigen formats
02
Fusion & Screening
Week 5–6
Electrofusion platform
ELISA-based screening
Hit identification
03
Subcloning & QC
Week 6–8
Subcloning to monoclonality
Isotype determination
Stability confirmation
04
Delivery
Week 8–9
Frozen hybridoma cells
Purified antibody (1–2 mg)
Validation data + COA
| Milestone | Timeline | Deliverables | Client Gate |
|---|---|---|---|
| M1: Immunization | Week 1–4 | Serum titer check (ELISA); immunization progress report | ✓ Approve |
| M2: Fusion & Hits | Week 5–6 | Primary ELISA hits; clone count; shortlist for subcloning | ✓ Select |
| M3: Subcloning | Week 6–8 | Monoclonal clones confirmed; isotype report; supernatant samples | ✓ Confirm |
| M4: Delivery | Week 8–9 | Frozen cells (≥5 vials) + purified Ab (1–2 mg) + ELISA data + COA + final report | ★ Complete |
Not sure if hybridoma is the right fit? Compare it with Single B Cell (45 days) and Phage Display (VHH/Nanobody) to find the best match for your target and application.
Explore real-world yeast protein expression case studies from AtaGenix. Our Pichia pastoris and Saccharomyces cerevisiae platforms deliver high-yield recombinant proteins with proper folding and glycosylation — supporting enzyme production, diagnostic antigen development, and antibody screening applications.
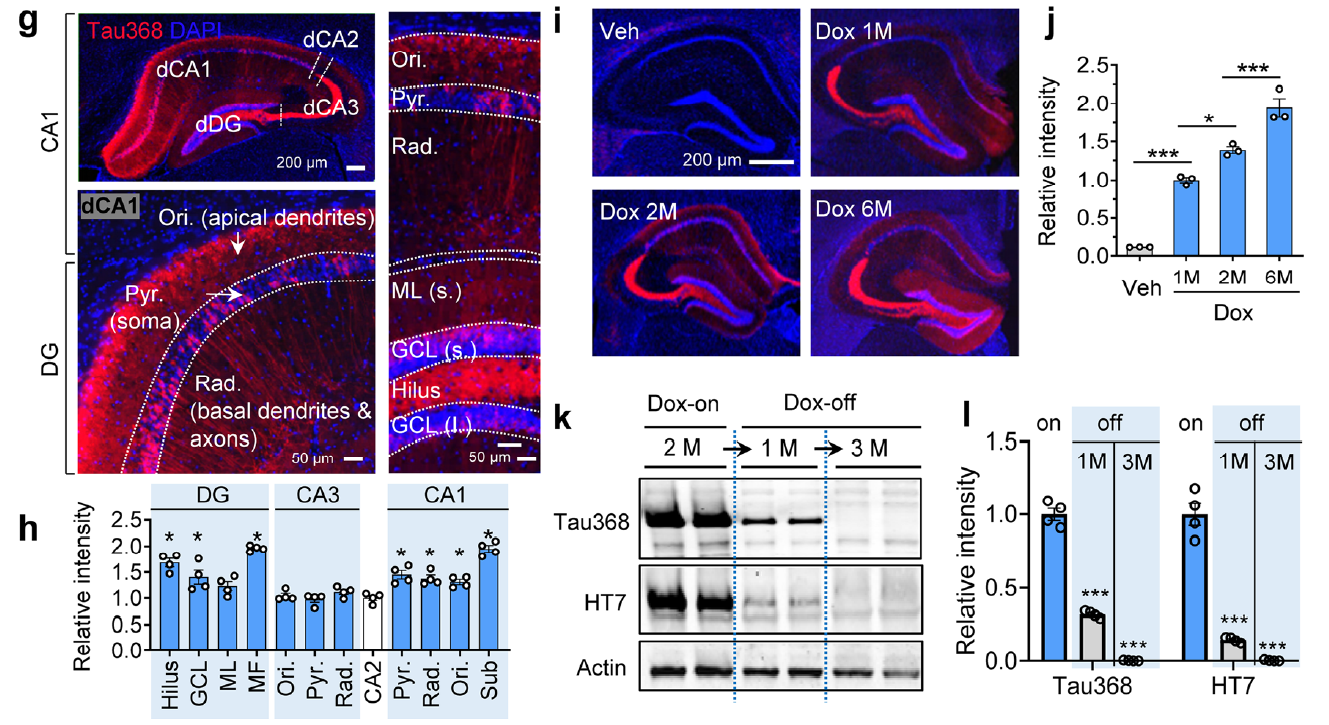
AtaGenix provides highly specific anti-tauN368 monoclonal antibodies to support Alzheimer’s disease research using the hTau368 transgenic mouse model. These antibodies enable precise detection of truncated tau fragments in Western blot and immunofluorescence experiments, validating tau accumulation, phosphorylation, and associated cognitive deficits in the hippocampus. With exceptional specificity and stability, AtaGenix’s solutions empower researchers to explore tau pathology mechanisms and advance tau-targeted therapeutic development.