AtaGenix Laboratories
AtaGenix Laboratories
AtaGenix provides Bacillus subtilis protein expression services for targets that require endotoxin-free production and direct secretion into culture medium. As a Gram-positive, GRAS-status organism, B. subtilis eliminates the endotoxin contamination inherent to E. coli systems — making it the preferred host for diagnostic reagents, food-grade enzymes, and proteins destined for in vivo applications.
Zero
Endotoxin by Nature
Secreted
Direct to Medium
GRAS
Regulatory Status
4 Strains
Host Library
The key advantage of B. subtilis is secretory expression: your target protein is exported directly into the culture supernatant via an efficient signal peptide and molecular chaperone system, bypassing cell lysis and dramatically simplifying downstream purification. For targets that require intracellular expression, we also support cytoplasmic production in dedicated strains.
| Expression Mode | Host Strains | Vectors |
| Secreted (extracellular) | WB800N | pHT01, pHT43, pHT254 |
| Intracellular (cytoplasmic) | AS1, 1012, BS168 |
| Factor | B. subtilis | E. coli |
| Endotoxin | Naturally endotoxin-free | Requires removal (<0.1 EU/µg) |
| Protein Location | Secreted to medium (or intracellular) | Cytoplasmic or inclusion body |
| Purification | Simplified (no lysis for secreted) | Cell lysis + refolding if IB |
| Inclusion Bodies | Avoided via signal peptide + chaperone system | Common for complex proteins |
| Best For | Diagnostics, food enzymes, in vivo | Lowest cost, intracellular targets |
Endotoxin-Free by Design
B. subtilis is Gram-positive — no LPS in the cell wall, no endotoxin removal steps needed. Your protein comes off the column clean enough for cell-based assays and in vivo studies without additional processing.
Secretion + Chaperone System
B. subtilis has an efficient signal peptide and molecular chaperone system that drives high-efficiency secretory expression, avoiding inclusion body formation. Target protein goes directly into the supernatant — no cell lysis, no refolding.
Dual Expression Modes
Secreted expression in WB800N for simplified purification, or intracellular expression in AS1/1012/BS168 when cytoplasmic production is preferred. Multiple vectors (pHT01, pHT43, pHT254) cover different promoter and cloning strategies.
AtaGenix's B. subtilis expression service covers the full pipeline from gene synthesis through purified protein delivery. Codon-optimized genes are cloned into B. subtilis-specific vectors (pHT01, pHT43, or pHT254), transformed into appropriate host strains (WB800N for secreted expression; AS1, 1012, or BS168 for intracellular), and screened for optimal expression. The best-performing clone is scaled to 1 L expression, followed by purification and comprehensive QC. B. subtilis is naturally endotoxin-free, so no LPS removal step is needed. Typical project timeline is 7–12 weeks from gene sequence to delivered protein.
A 4-stage pipeline with expression screening built in. Typical turnaround: 7–12 weeks from gene sequence to delivered protein.
01
Gene Synthesis
2–4 weeks
Codon optimization
Vector cloning (pHT01/43/254)
Sequence verification
02
Expression Screening
~2 weeks
Strain transformation
WB800N / AS1 / 1012 / BS168
Expression & solubility test
03
1 L Expression & Purification
~2 weeks
Scale to 1 L culture
Supernatant capture (secreted)
or cell lysis (intracellular)
Chromatography purification
04
QC & Delivery
Included in Stage 3
SDS-PAGE, Western Blot
Concentration analysis
Project report + protein sample
Optional Scale-Up: After the 1 L expression stage, larger-scale fermentation and purification (1–4 additional weeks) is available upon request for projects requiring higher quantities.
Service Scope
| ✓ B. subtilis codon optimization & gene synthesis | ✓ Vectors: pHT01, pHT43, pHT254 |
| ✓ Secreted expression host: WB800N | ✓ Intracellular hosts: AS1, 1012, BS168 |
| ✓ High-efficiency competent cell preparation | ✓ Signal peptide & chaperone-assisted secretion |
| ✓ 1 L expression + optional fermentation scale-up | ✓ Chromatography purification |
| ✓ QC: SDS-PAGE, Western Blot, concentration | ✓ Naturally endotoxin-free (no LPS removal) |
| Use Case | Why B. subtilis |
| Diagnostic reagent antigens | Endotoxin-free production eliminates false positives in cell-based diagnostic assays |
| Industrial enzymes | GRAS status + high secretion yield = food-grade and industrial-grade production |
| Proteins for in vivo studies | No endotoxin removal step needed, reducing processing time and cost |
| Secreted protein targets | Signal peptide + chaperone system avoids inclusion body formation, yields soluble protein |
Need a different expression system? Compare with E. coli for lowest-cost intracellular production, HEK293 for human-type glycosylation, or CHO Transient for therapeutic antibody leads.
What types of proteins work best in B. subtilis?
B. subtilis excels at producing secreted proteins, particularly enzymes, small-to-medium soluble proteins, and antigens for diagnostic applications. The built-in signal peptide and chaperone system helps avoid inclusion body formation that is common in E. coli. Proteins requiring complex eukaryotic glycosylation or disulfide-heavy structures are better suited to mammalian expression systems.
Is B. subtilis expression truly endotoxin-free?
Yes. B. subtilis is a Gram-positive bacterium and does not produce lipopolysaccharide (LPS). This is a biological property of the organism, not a purification step — endotoxin levels are intrinsically undetectable rather than reduced post-production.
Can B. subtilis do both secreted and intracellular expression?
Yes. We offer dedicated strains for each mode: WB800N for secreted expression (protein goes directly into culture medium), and AS1, 1012, or BS168 for intracellular (cytoplasmic) expression. The choice depends on your target protein's properties and downstream application. Our team will recommend the optimal strain and vector combination during project consultation.
What is the typical timeline and what QC data is provided?
A standard project takes 7–12 weeks: gene synthesis (2–4 weeks), expression screening (2 weeks), 1 L expression and purification (2 weeks), plus optional larger-scale fermentation (1–4 weeks). Every project includes SDS-PAGE, Western Blot, protein concentration analysis, and a detailed technical report. Additional QC is available upon request.
Timelines and yields depend on target protein properties and secretion efficiency. Quote-based pricing.
Explore real-world yeast protein expression case studies from AtaGenix. Our Pichia pastoris and Saccharomyces cerevisiae platforms deliver high-yield recombinant proteins with proper folding and glycosylation — supporting enzyme production, diagnostic antigen development, and antibody screening applications.
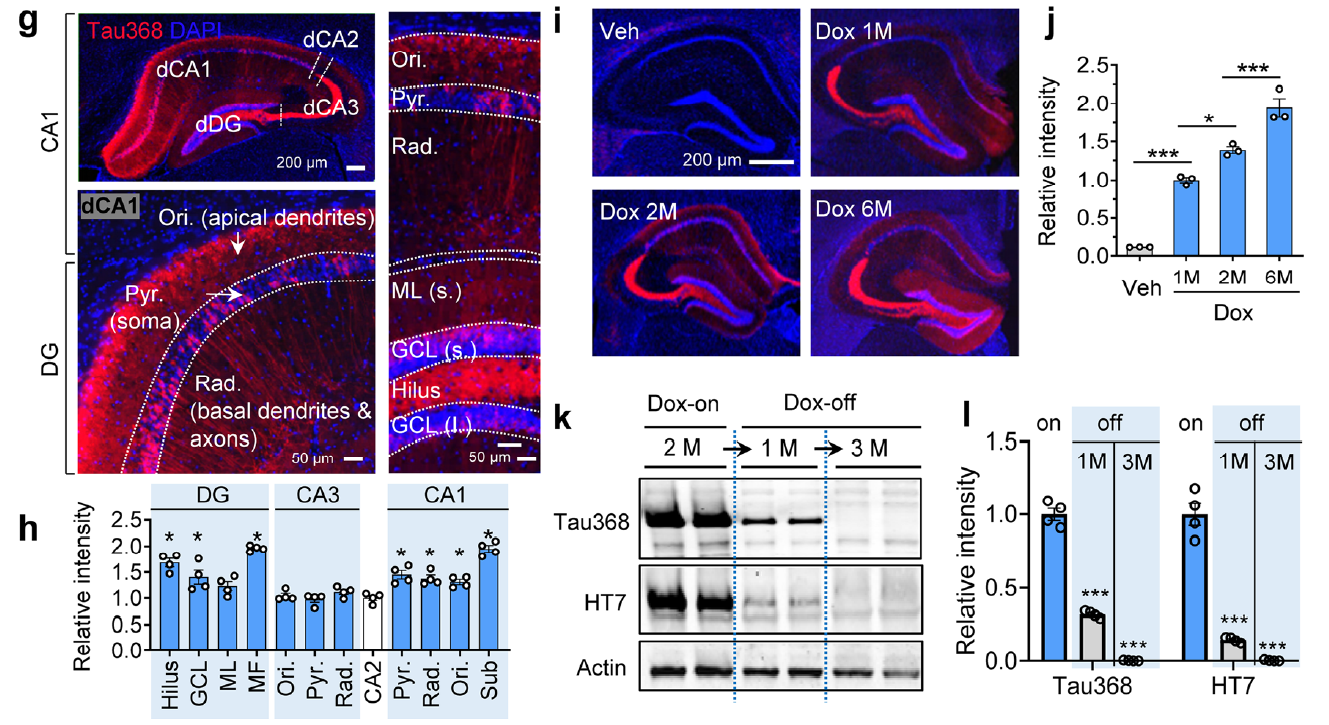
AtaGenix provides highly specific anti-tauN368 monoclonal antibodies to support Alzheimer’s disease research using the hTau368 transgenic mouse model. These antibodies enable precise detection of truncated tau fragments in Western blot and immunofluorescence experiments, validating tau accumulation, phosphorylation, and associated cognitive deficits in the hippocampus. With exceptional specificity and stability, AtaGenix’s solutions empower researchers to explore tau pathology mechanisms and advance tau-targeted therapeutic development.