AtaGenix Laboratories
AtaGenix Laboratories
AtaGenix's mammalian protein expression platform offers both transient and stable production routes using CHO and HEK293 host cells. Mammalian expression ensures native post-translational modifications — including complex N-linked glycosylation, disulfide bond formation, and proper folding — critical for therapeutic antibodies, Fc-fusion proteins, receptor ectodomains, and viral antigens.
>7 g/L
CHO Stable Titer
~2 wks
Transient Delivery
XtenCHO™
Proprietary High-Density System
200L
Scale-Up Capacity
Our proprietary XtenCHO™ high-density expression system is specifically optimized for difficult-to-express proteins, delivering higher titers and faster development timelines compared to standard CHO platforms. Choose transient expression for rapid milligram-scale delivery (screening, SPR, animal studies), or stable cell line development for consistent gram-scale supply (preclinical, manufacturing).
| Service | Best For | Timeline | Scale |
| CHO Transient | Manufacturing-representative glycosylation; XtenCHO™ for difficult proteins; therapeutic Ab screening | ~2 weeks | mg–100s mg |
| HEK293 Transient | Human-type glycosylation; gene therapy proteins; viral antigens; receptor ectodomains; rapid turnaround | ~2 weeks | mg–100s mg |
| CHO Stable Cell Line | Long-term gram-scale supply; manufacturing-ready; clonal cell line; Research Cell Bank; GS/DHFR selection | 4–6 months | g–multi-g |
| Factor | CHO | HEK293 |
| Glycosylation | Manufacturing-representative (matches commercial biologic production) | Human-type (higher sialylation, bisecting GlcNAc) |
| Best For Proteins | Therapeutic antibodies, Fc-fusions, biosimilars | Viral antigens, gene therapy proteins, receptor ectodomains, complex multi-subunit |
| Stable Cell Line | Available (GS/DHFR selection, >7 g/L fed-batch) | Possible but CHO preferred for manufacturing |
| Regulatory Path | Preferred for IND/BLA (most approved biologics use CHO) | Accepted for gene therapy and some biologics |
| Transfection Efficiency | Good (XtenCHO™ optimized) | Excellent (high transfection efficiency, fast expression) |
XtenCHO™ Proprietary System
Our high-density CHO transient system delivers consistently higher titers than standard CHO platforms, especially for difficult-to-express proteins. Optimized media, transfection protocol, and cell line enable mg-scale production in just 2 weeks.
Transient-to-Stable Upgrade Path
Start with transient expression to validate your protein, then seamlessly transition to CHO stable cell line development when you need gram-scale supply. Same vector backbone, same expression system — no re-optimization needed.
Scale-Up to 200L
From transient milligram-scale to 200L bioreactor production. Fed-batch optimization, process parameter transfer, and full QC documentation for preclinical and early clinical material supply.
Explore real-world yeast protein expression case studies from AtaGenix. Our Pichia pastoris and Saccharomyces cerevisiae platforms deliver high-yield recombinant proteins with proper folding and glycosylation — supporting enzyme production, diagnostic antigen development, and antibody screening applications.
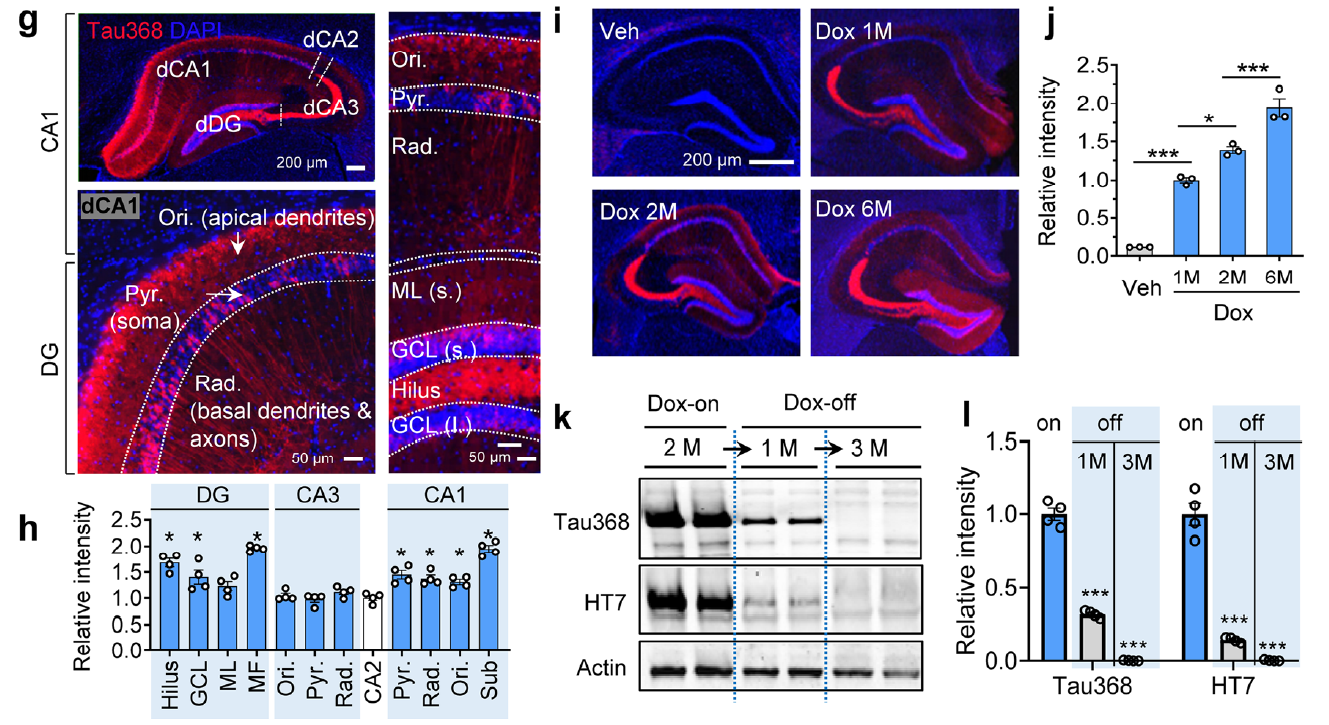
AtaGenix provides highly specific anti-tauN368 monoclonal antibodies to support Alzheimer’s disease research using the hTau368 transgenic mouse model. These antibodies enable precise detection of truncated tau fragments in Western blot and immunofluorescence experiments, validating tau accumulation, phosphorylation, and associated cognitive deficits in the hippocampus. With exceptional specificity and stability, AtaGenix’s solutions empower researchers to explore tau pathology mechanisms and advance tau-targeted therapeutic development.