AtaGenix Laboratories
AtaGenix Laboratories
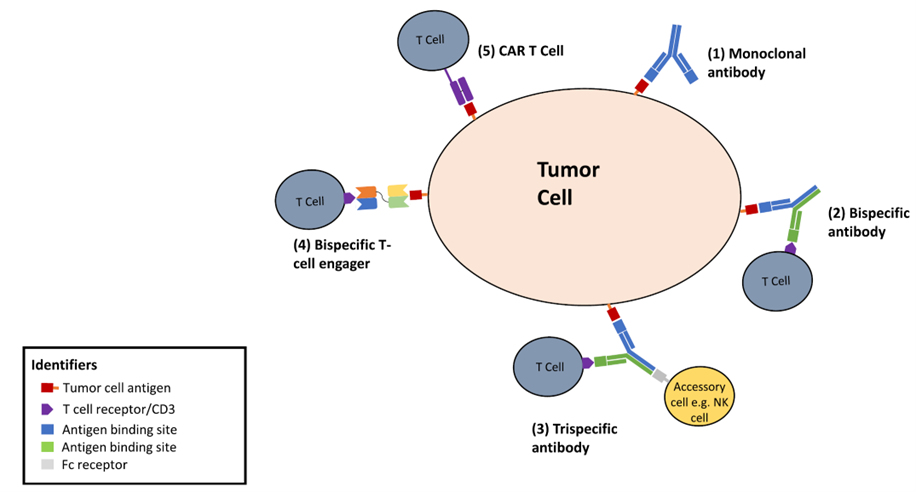
Bispecific antibodies (BsAbs) and trispecific antibodies (TsAbs) represent a significant evolution in cancer immunotherapy. By simultaneously targeting two or three antigens, these multispecific antibodies enable precise immune cell redirection, overcome tumor heterogeneity and antigen escape, and enhance anti-tumor efficacy. With over 17 BsAbs now approved—primarily for hematologic malignancies—and trispecific candidates showing promising early clinical results (such as high response rates in relapsed/refractory multiple myeloma with favorable safety), this field is rapidly advancing. This article reviews their mechanisms, key clinical candidates, current challenges, and future prospects in both blood cancers and solid tumors.
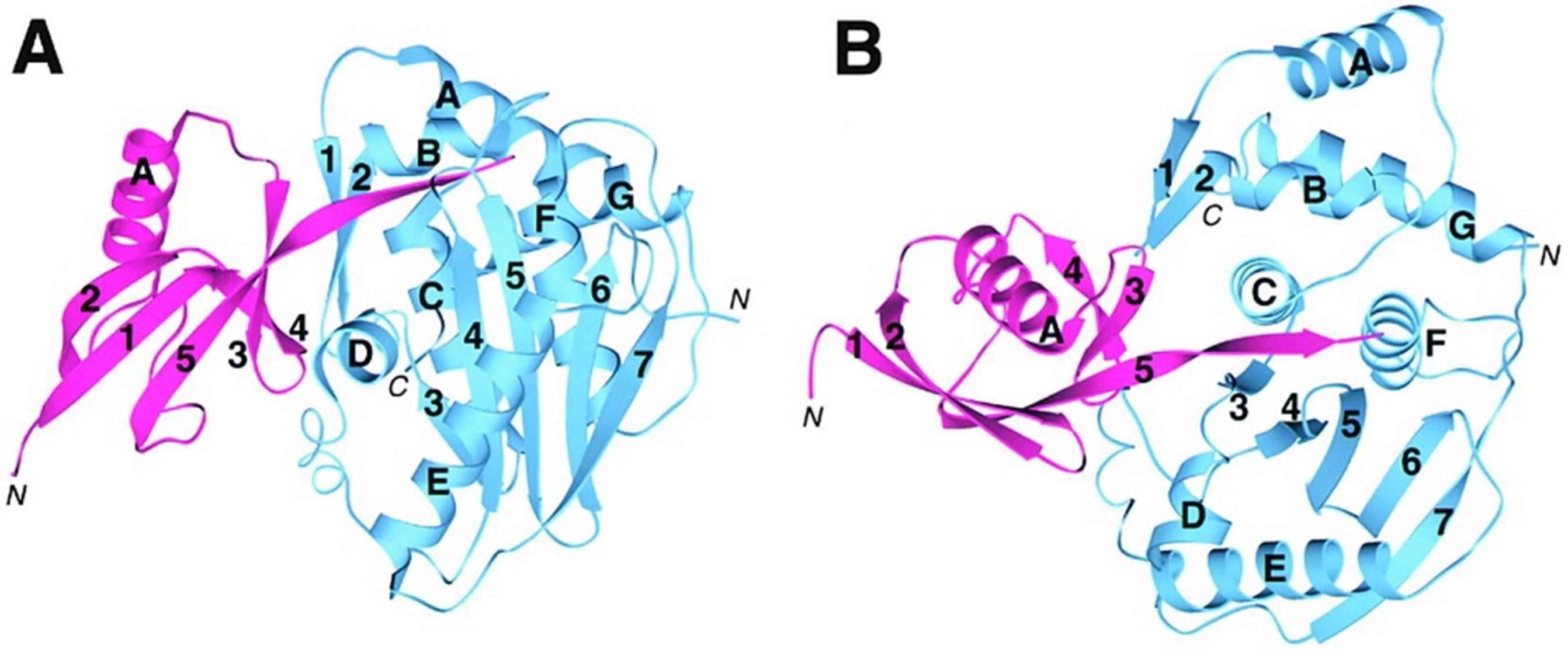
Affinity tags such as His, GST, MBP, and SUMO significantly enhance the yield and solubility of recombinant proteins during expression and purification. However, these tags frequently interfere with the target protein’s structure, function, or downstream applications, including crystallization, activity assays, and drug development. Precise removal of the tag has therefore become a critical step in obtaining native-like proteins.
Enzymatic cleavage remains the most reliable method due to its superior specificity compared to chemical cleavage. This article reviews commonly used endoproteases and exoproteases, focusing on their advantages and limitations. Special emphasis is placed on viral cysteine proteases — TEV, HRV 3C (PreScission™), and SUMO proteases (Ulp1/SENP family) — which belong to the clan CA cysteine protease superfamily.
The catalytic mechanisms, substrate recognition modes (sequence-specific vs. structure-specific), detergent tolerance, and optimized variants (including S219V TEV, SUMOstar, and the novel consensus-designed Con1 protease) are discussed in detail. Practical considerations for membrane protein purification and strategies for achieving scarless or near-native N-termini are also highlighted.
By integrating solubility tag design with highly specific enzymatic cleavage tools, researchers can efficiently produce high-quality, tag-free recombinant proteins suitable for structural biology, functional studies, and therapeutic development.
Comprehensive overview of therapeutic antibody derivatives: ADCs (10 FDA-approved, 80+ in clinical trials), ARCs (antibody-oligonucleotide conjugates), bispecific/multispecific antibodies (BiTE, CrossMab, Knobs-into-Holes), immunocytokines (IL-2, IL-12, TGF-β trap fusions), and antibody fragments (Fab, scFv, VHH nanobodies, DARPins). Covers mechanisms, approved products, and future directions including TCR-mimic antibodies for intracellular antigen targeting.

AtaGenix developed a custom phospho-specific antibody against human USP8 Ser718, enabling an Advanced Science (IF 14.3) study that identified USP8 as the core deubiquitinase governing MDA5 homeostasis. The AKT–USP8–MDA5 axis drives type I interferon signaling; inhibiting USP8 or AKT suppresses MDA5-induced autoimmunity in AGS mice and anti-MDA5-positive DM/SLE patient cells. DOI: 10.1002/advs.202503865.
A Cancer Communications study mapped the pAKT–pPCK1–pLDHA–SPRINGlac axis as a driver of ferroptosis and chemo-immunotherapy resistance in AKT-hyperactivated intrahepatic cholangiocarcinoma. Simvastatin disrupted mevalonate flux and restored therapy sensitivity in preclinical models. pAKT–pPCK1 emerged as a biomarker for patient stratification. AtaGenix provided custom phospho-specific antibodies (anti-pPCK1 Ser90, anti-pLDHA Thr248) validated for WB/IP/IHC. DOI: 10.1002/cac2.70036.
An Advanced Science study (IF 14.3) identified the PAD1–AKT2(R202)–CEBPβ axis as a driver of ovarian cancer stem-like cell stemness and cisplatin resistance. PAD1 citrullinates AKT2 at Arg202, maintaining phosphorylation site exposure and activating PI3K-AKT–CEBPβ signaling. PAD1 inhibition re-sensitizes cisplatin-resistant cells. AtaGenix provided a custom site-specific Cit202 antibody validated for co-IP and IHC across 66 clinical samples. DOI: 10.1002/advs.202501014.

AtaGenix provides end-to-end bispecific antibody custom development supporting 15+ formats (BiTE, IgG-like, Fab-based, nanobody-based, multispecific). >90% project success rate using proprietary HEK293 and XtenCHO™ expression platforms. Full workflow from gene synthesis and vector construction to expression, purification, and QC (SDS-PAGE, SEC-HPLC, ELISA) in 5–6 weeks. Case studies include anti-EBV gH/gL and anti-GPC3×CD3E bispecific antibodies.

AtaGenix's BEVS platform (Sf9/Sf21/Hi5) delivers properly folded, glycosylated recombinant proteins from milligram to gram scale. Four peer-reviewed publications showcase BEVS-produced reagents in oncology (Nat Commun, POLD1-MYC), tumor immunology (Biomed Pharmacother, PADI4 mAb), neuroscience (Nat Commun, PCIF1-MTase), and gynecological oncology (AJCR, ENO1 mAb). ISO 9001 & ISO 13485 certified.

AtaGenix's 3H E. coli expression platform (>95% success rate) delivers publication-grade recombinant proteins from mg to gram scale. Optimized vectors (pET/pGEX), specialized strains (T7E, C41, Arctic), and full QC (purity ≥85%, endotoxin <0.1 EU/mL). Three peer-reviewed case studies — all in Nature Communications — showcase E. coli-expressed reagents for rice genetics (SDR3.1), Bt resistance (FTZ-F1), and cyanobacterial stress tolerance (shikimate kinase).
AtaGenix's mammalian expression platform (HEK293/CHO-S transient, CHO-K1/DG44 stable) has delivered 5,000+ projects with 200+ stable cell lines. Four peer-reviewed publications showcase mammalian-expressed reagents: OMV tumor vaccine (Adv Mater IF 27.4), SARS-CoV-2 nanovaccine (Nat Commun IF 14.7), EAE neuroinflammation (Immunity IF 25.5), and PND complement inhibition (Mol Psychiatry IF 9.6). Class 100,000 cleanroom, endotoxin <0.1 EU/mL, ISO certified.
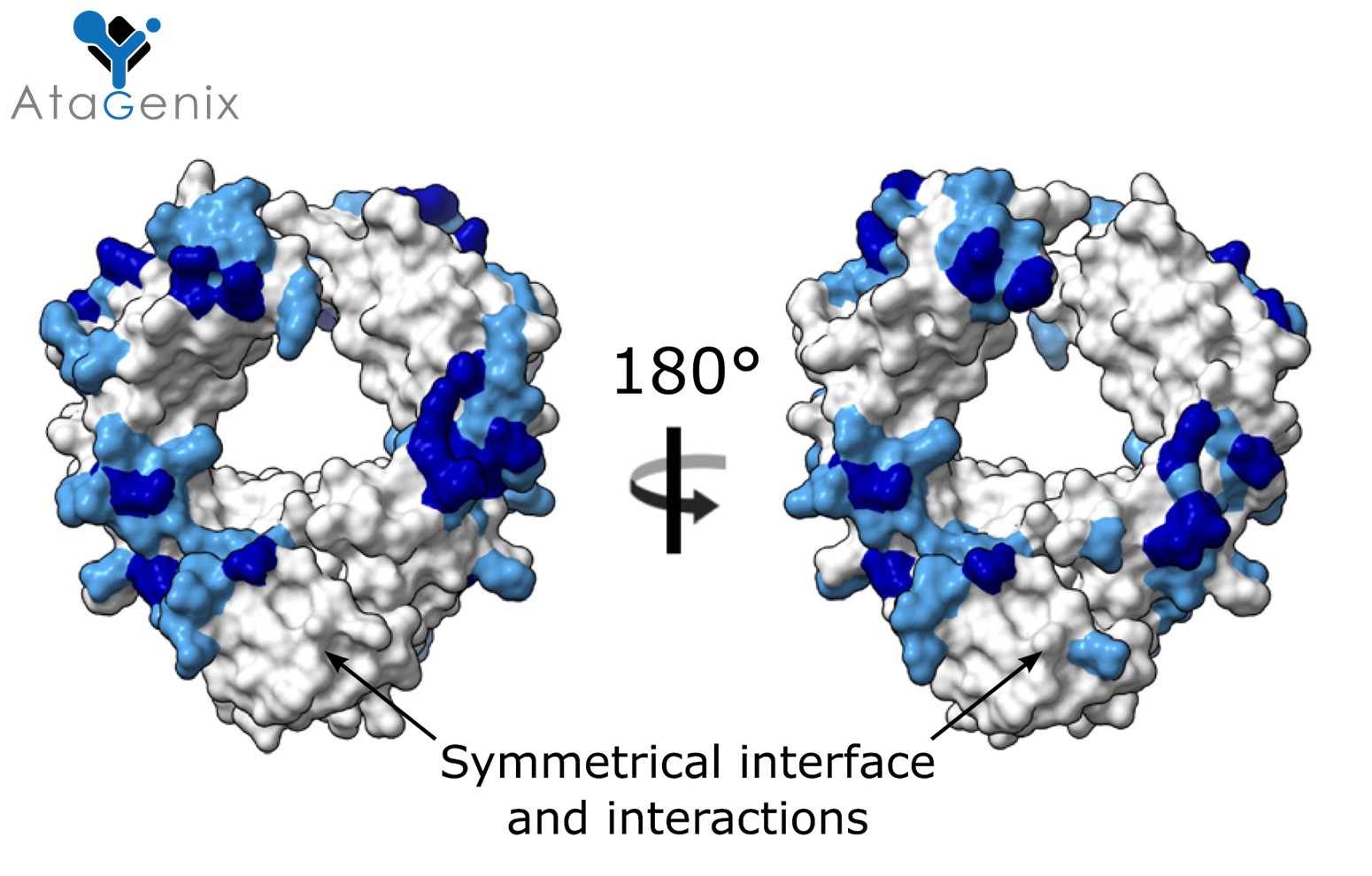
A Nature Communications study resolved the crystal structures of IdeS–IgG1 Fc and EndoS–IgG1 Fc complexes, revealing the molecular basis of substrate-specific recognition. IdeS cleaves the IgG hinge region; EndoS hydrolyzes the Fc N-glycan. Clinical applications include Guillain-Barré syndrome (37% independent walking at week 1), AAV gene therapy antibody clearance (Nature Medicine 2020), and anti-GBM renal disease (GOOD-IDES Phase II). AtaGenix offers recombinant IdeS protease (ATE00010, >95% purity, 40 U/μL).
A Nature Microbiology study revealed that the E2 enzyme in E2-CBASS regulates cGAS via ubiquitination mimicry, promoting poly-cGAS formation (4.2× increase) and cGAMP synthesis (2.3× boost) to confer anti-phage immunity. Cryo-EM resolved the cGAS-E2 covalent complex. AtaGenix provided a custom rabbit anti-cGAS polyclonal antibody that distinguished cGAS monomers (46 kDa), complexes (64 kDa), and poly-cGAS (>100 kDa) in Western blot. DOI: 10.1038/s41564-024-01684-z.
Contact Us
+86-27-6552-3339
info@atagenix.com
Building C, R & D Building, No. 666, Shendun 4th Road, Donghu New Technology Development Zone, Wuhan

