AtaGenix Laboratories
AtaGenix Laboratories
Release time: 2025-08-28 View volume: 1133
Project Snapshot — Leveraging high-purity recombinant mouse MOG protein provided by AtaGenix, researchers established an EAE model and uncovered how meningeal B cells drive multiple sclerosis (MS) relapse through MHC II-mediated antigen presentation, GM-CSF–dependent neutrophil recruitment, and endothelial activation. Local intracisternal anti-CD20 selectively depleted brain-resident B cells and significantly reduced relapse severity — published in Immunity (IF: 25.5).
Based on: Immunity, 2025 — DOI: 10.1016/j.immuni.2025.06.016 (IF: 25.5, Q1)
Multiple sclerosis relapses are driven by incompletely understood CNS-intrinsic immune mechanisms. While peripheral B-cell depletion (anti-CD20) is effective in relapsing–remitting MS, the specific role of brain-resident meningeal B cells remained unclear. This study used a custom-produced MOG protein from AtaGenix as the core autoantigen to build a robust EAE model, enabling precise dissection of meningeal B-cell pathogenicity.
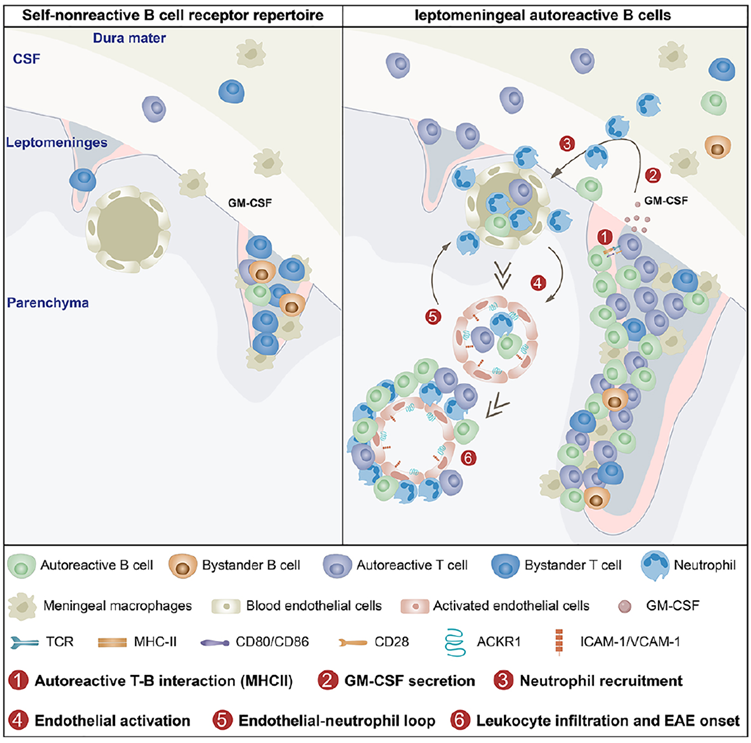
Fig. 1. Graphical abstract: meningeal B cells drive neuroinflammatory relapse through antigen presentation and a self-sustaining inflammatory loop. Adapted from Immunity 2025.
Under normal conditions, meningeal B cells maintain strict immune tolerance. However, when autoreactive B cells targeting CNS antigens were introduced, they became specifically activated within the leptomeninges and acquired antigen-presenting capacity. This activation was strictly dependent on the presence of CNS autoantigens — mice lacking MOG failed to trigger B-cell activation, confirming MOG’s central role in driving this pathological process. The high-purity, biologically active MOG protein used throughout these experiments was provided by AtaGenix, ensuring model consistency and reliable data.
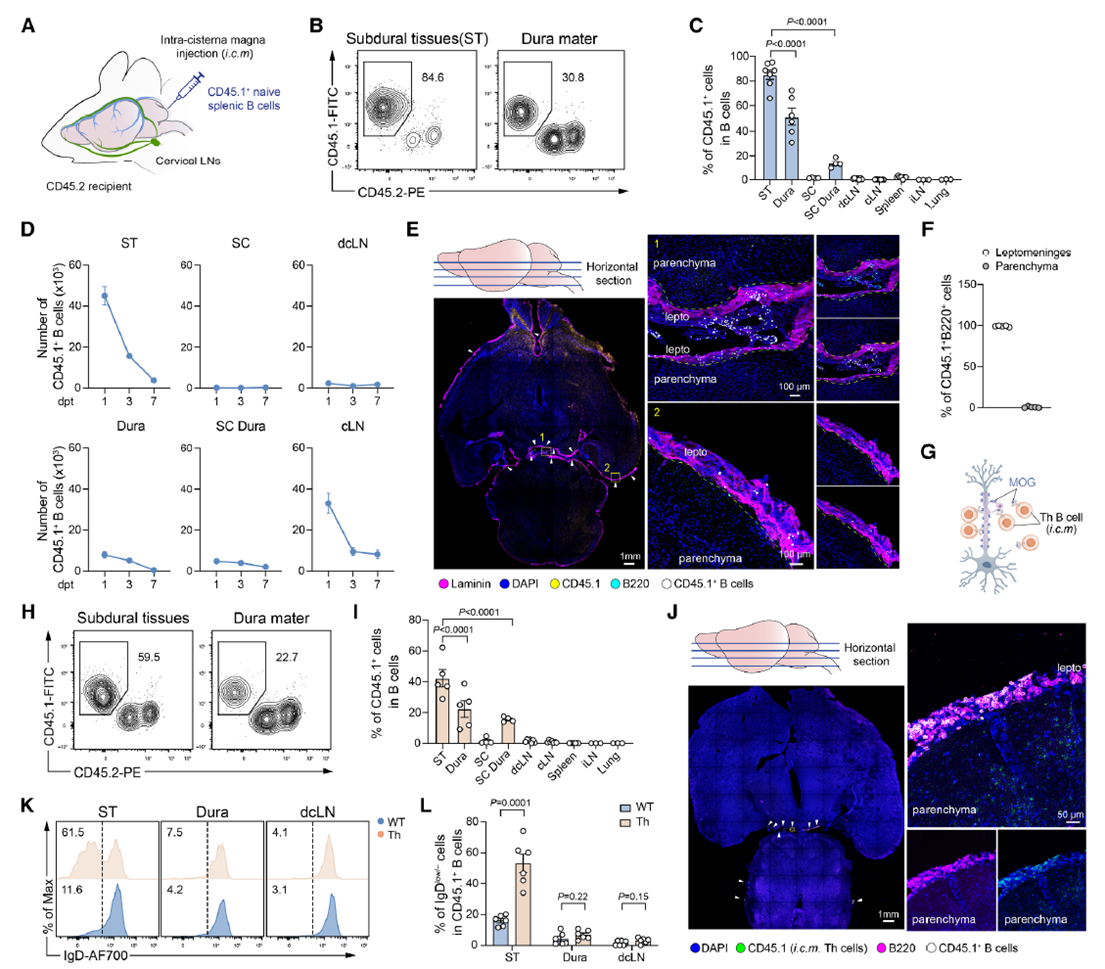
Fig. 2. Meningeal autoreactive B cells promote T cell-mediated CNS inflammation through antigen presentation. AtaGenix-produced MOG protein served as the core autoantigen. Adapted from Immunity 2025.
Autoreactive meningeal B cells present MOG antigens to pathogenic T cells via MHC II molecules, triggering rapid T-cell activation and robust GM-CSF production. GM-CSF in turn recruited neutrophils into the meninges, amplifying the inflammatory response. Once present, neutrophils activated vascular endothelial cells, inducing adhesion molecules (VCAM-1, ACKR1) that facilitated further immune-cell infiltration — creating a self-sustaining B cell → T cell → neutrophil → endothelium inflammatory loop.
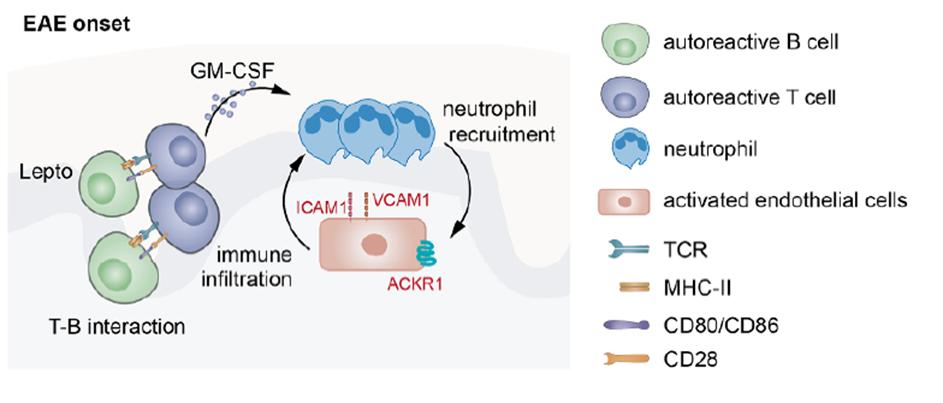
Fig. 3. A proinflammatory loop between GM-CSF–dependent neutrophil recruitment and endothelial cell activation. Adapted from Immunity 2025.
In a relapsing–remitting EAE model, local intracisternal injection of low-dose anti-CD20 selectively depleted brain-resident B cells without affecting peripheral B-cell populations. This intervention significantly reduced relapse severity, suppressed T-cell proliferation, lowered pro-inflammatory cytokine release, and diminished endothelial activation. Single-cell sequencing further revealed substantial clonal expansion of brain B cells during relapse that persisted into remission, highlighting their role as both initiators and drivers of recurrence.
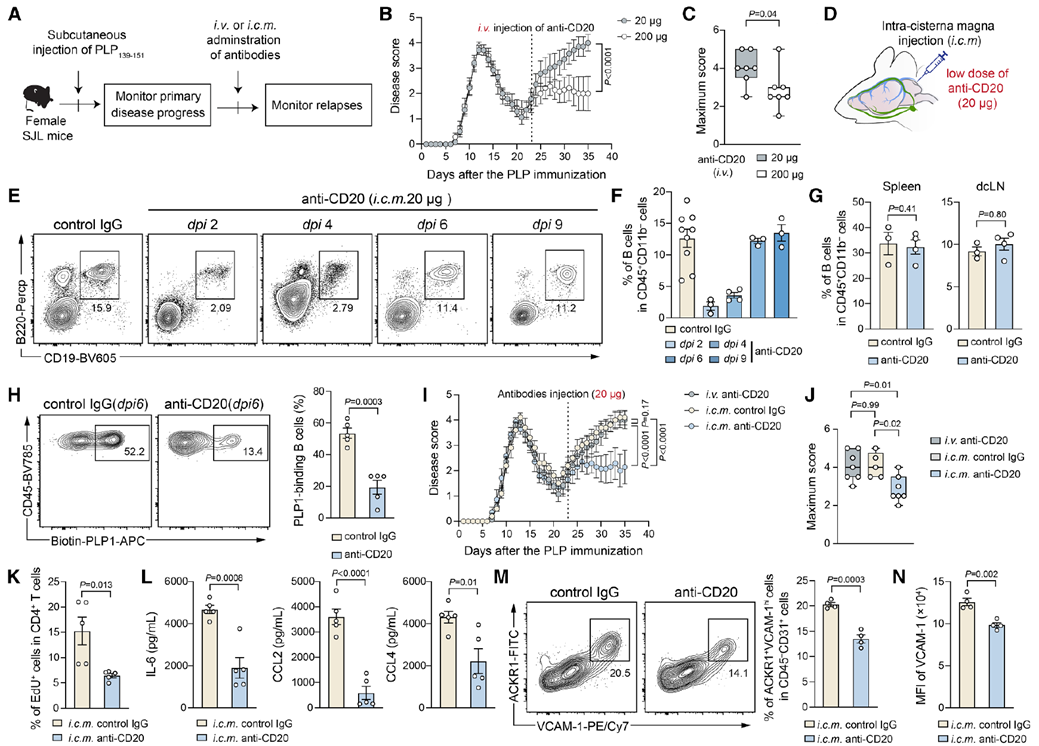
Fig. 4. Targeted depletion of brain-localized B cells reduces EAE relapse. Local anti-CD20 selectively removed meningeal B cells without peripheral effects. Adapted from Immunity 2025.
25.5
Impact Factor (Immunity)
MOG
Core Autoantigen Supplied
EAE
Model Established
anti-CD20
Local Therapy Validated
Why This Matters
This study fills a critical gap in understanding how CNS-resident B cells initiate and sustain MS relapses independently of peripheral immune responses. The AtaGenix-produced recombinant MOG protein was central to establishing the model — MOG-knockout mice failed to activate meningeal B cells, confirming that the antigen’s quality directly determined experimental validity. The finding that local anti-CD20 can mitigate relapse without systemic immunosuppression opens a new avenue for targeted MS therapy, with implications for anti-CD20 and CD40 inhibitor development.
Results may vary depending on target protein, model design, and project scope. All proprietary client information is subject to NDA.
Need high-purity recombinant autoantigens for EAE, MS, or other autoimmune disease models? AtaGenix delivers publication-grade proteins with native conformation via mammalian cell expression.
Talk to Technical SupportResponse within 24 hours
Contact Us
+86-27-6552-3339
info@atagenix.com
Building C, R & D Building, No. 666, Shendun 4th Road, Donghu New Technology Development Zone, Wuhan

