AtaGenix Laboratories
AtaGenix Laboratories
Release time: 2025-09-02 View volume: 1430
News Highlight — The bispecific antibody–drug conjugate (BsADC) pipeline has surpassed 100 clinical candidates, with China contributing 60+ programs. Industry analysis projects the first commercial BsADC approval around 2029 — signaling a pivotal inflection point for multispecific biologics and targeted oncology.
Sources: Nature (industry/technology overview) · Clinical Trial Vanguard
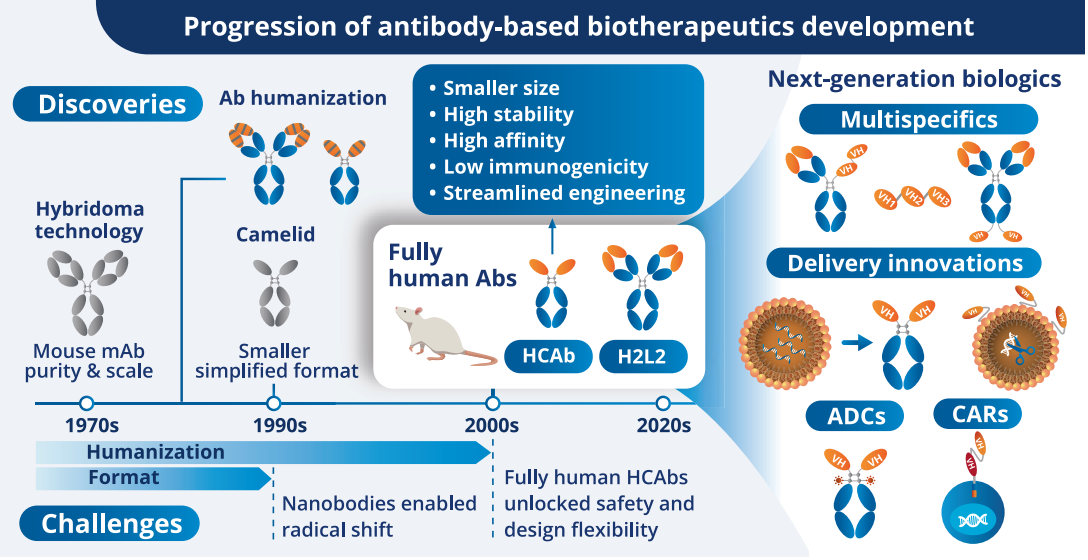
Progression of antibody-based biotherapeutics development: hybridoma-derived mAbs → humanization → next-generation modalities (multispecifics, ADCs, CARs). Adapted from cited literature.
100+
Clinical BsADC Programs
60+
China-Originated Programs
~2029
Projected First Approval
Ph 2/3
Multiple Readouts Expected
Modalities Converge
Targeting flexibility: Bispecific scaffolds enable dual-antigen recognition, improving tumor selectivity over monospecific ADCs.
Payload innovation: New linker–payload systems (topoisomerase I inhibitors, MMAE/MMAF, site-specific conjugation) broaden therapeutic windows.
Manufacturability: Advances in developability engineering mitigate aggregation, PK liabilities, and DAR heterogeneity.
Pipeline Depth & Geography
Global scale: 100+ clinical BsADC programs across oncology, with multiple Phase 2/3 readouts expected in the near term.
China leadership: 60+ programs reflect strong domestic innovation and capital, complementing North America/EU portfolios.
Regulatory momentum: Experience from mAb and ADC approvals is informing BsADC CMC and clinical development strategies.
| Axis | Current Emphasis |
|---|---|
| Targets | Tumor antigens (HER2, TROP2, EGFR, Claudin family) combined with internalization-favorable pairs |
| Linker / Payload | Cleavable linkers; topoisomerase I inhibitors, MMAE/MMAF; site-specific conjugation for DAR homogeneity |
| Format | Symmetric/asymmetric IgG-like, knob-into-hole, tandem scFv; Fc engineering for PK/effector tuning |
| Clinical Readouts | ORR/PFS improvements in biomarker-selected populations; safety optimization for combination regimens |
AtaGenix Perspective
With the BsADC pipeline approaching a commercialization inflection point, the engineering challenges for early-stage programs are substantial: bispecific format selection, chain-pairing optimization, linker–payload strategy, DAR control, and scalable manufacturing all must be addressed in parallel. AtaGenix provides integrated support across this workflow:
Bispecific Design & Engineering: 15+ formats supported, including humanization and developability screening (liability, aggregation risk assessment).
Antibody Discovery: Single B cell (Xten™ Mab), hybridoma, and phage display platforms for lead identification.
Multi-System Expression: E. coli, yeast, mammalian (HEK293/XtenCHO™), and insect cell (BEVS) for recombinant protein and antibody production.
Stable Cell Lines & Scale-Up: CHO-K1/DG44 with G418, DHFR, or GS selection for preclinical and manufacturing supply (~12 weeks to RCB).
Note: Pipeline counts (100+ BsADCs; China 60+) and projected timelines are synthesized from the cited sources; individual methodologies may vary.
Developing a bispecific ADC or multispecific program? AtaGenix supports the full journey from bispecific design to conjugation-ready antibody production.
Talk to Technical SupportResponse within 24 hours
Contact Us
+86-27-6552-3339
info@atagenix.com
Building C, R & D Building, No. 666, Shendun 4th Road, Donghu New Technology Development Zone, Wuhan

