AtaGenix Laboratories
AtaGenix Laboratories
Release time: 2025-08-28 View volume: 1128
Project Snapshot — With AtaGenix’s Pichia pastoris expression support, researchers produced recombinant Patatin (a potato-derived lipase) and applied rational site-directed mutagenesis to overcome its inherent short-chain substrate preference. The D286A mutant achieved a 3.2-fold increase in long-chain fatty acid (pNP-C16) specificity with improved thermal stability — opening new applications in functional lipid production, green biocatalysis, and food biotechnology.
Patatin is a potato glycoprotein with emulsifying, antioxidant, and lipid-modifying properties that make it attractive as a plant-derived lipase. However, two limitations hinder industrial adoption: low natural expression levels and a strong preference for short-chain substrates, leaving catalytic efficiency toward long-chain fatty acids relatively weak.
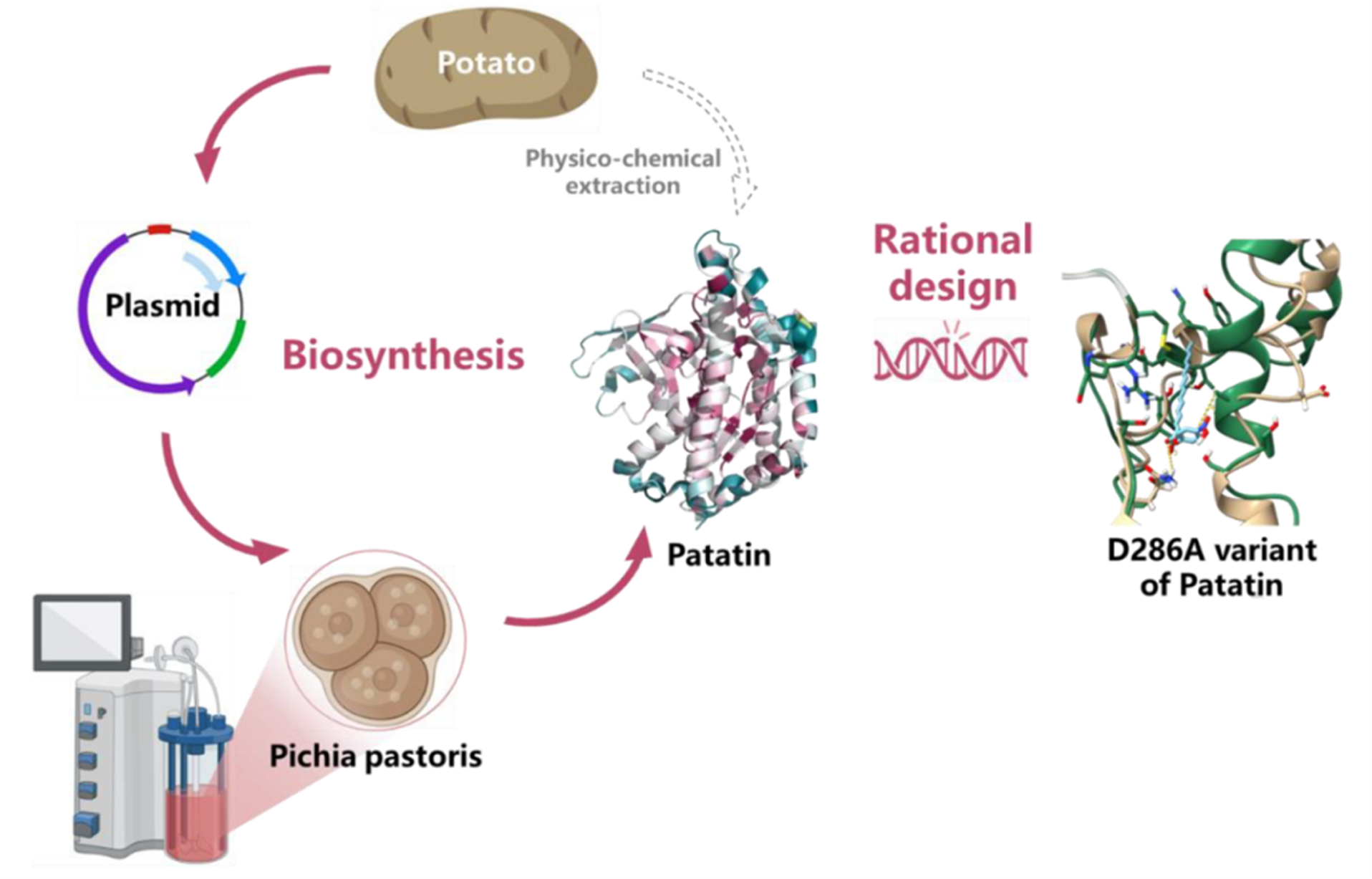
Fig. 1. Biosynthesis of the D286A mutant. Recombinant Patatin expressed in Pichia pastoris with AtaGenix support.
Using the Pichia pastoris expression system provided by AtaGenix, the team produced recombinant Patatin at sufficient yields (121 mg/L) for structural engineering and functional studies. Enzymatic characterization revealed an optimal temperature of ~35 °C, optimal pH of 10.0, and good alkaline stability. Activity was enhanced by methanol but strongly inhibited by Fe²+/Fe³+. While the wild-type enzyme showed high activity toward short-chain substrates (pNP-C4), catalytic efficiency for long-chain substrates (pNP-C16) remained limited.
To overcome the short-chain preference, the researchers targeted residues near the substrate-binding pocket via rational site-directed mutagenesis. The D286A mutant showed the most striking improvement:
Enhanced Specificity: Activity toward long-chain substrate pNP-C16 increased 3.2-fold over wild type.
Structural Adaptation: The D286A mutation disrupted a local α-helix and formed a flexible loop, improving accommodation of long-chain acyl groups.
Molecular Mechanism: MD simulations revealed strengthened hydrogen bonding and a new π-alkyl interaction that stabilized substrate binding.
Stability: Improved thermal stability; CD spectroscopy confirmed overall folding remained unchanged.
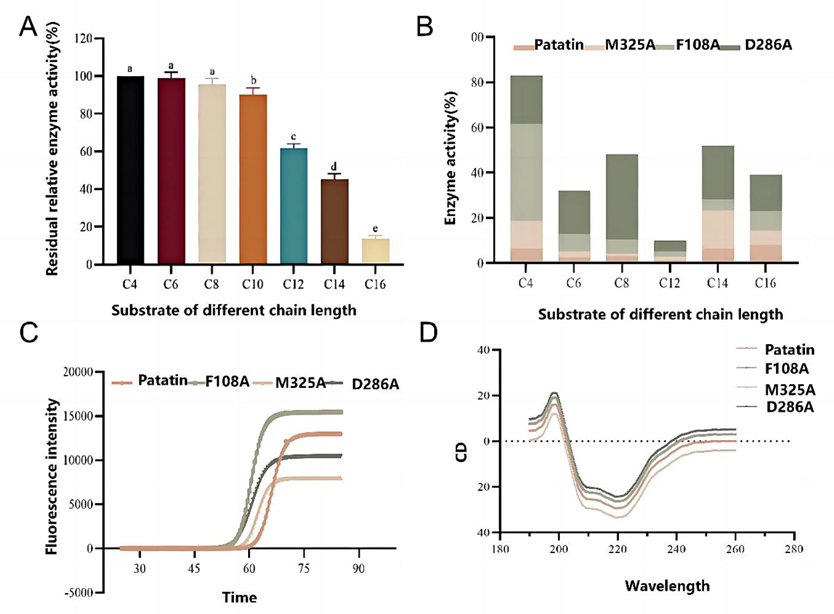
Fig. 2. Substrate specificity of lipase Patatin. D286A mutant shows 3.2-fold enhanced activity toward long-chain pNP-C16.
3.2×
Long-Chain Activity Gain
D286A
Optimized Variant
121
mg/L Expression Yield
P. pastoris
Expression System
Why This Matters
This is the first demonstration that rational design can effectively shift Patatin’s substrate preference from short-chain to long-chain fatty acids, addressing a long-standing bottleneck in plant-derived lipase applications. The D286A variant expands catalytic potential for functional lipid production and green biocatalysis, while the AtaGenix Pichia pastoris expression platform delivered the yields and quality needed for iterative protein engineering cycles.
Results may vary depending on target protein, construct design, and project scope. All proprietary client information is subject to NDA.
Need yeast expression for enzyme engineering, directed evolution, or industrial protein production? AtaGenix offers Pichia pastoris and Saccharomyces cerevisiae platforms with end-to-end optimization support.
Talk to Technical SupportResponse within 24 hours
Contact Us
+86-27-6552-3339
info@atagenix.com
Building C, R & D Building, No. 666, Shendun 4th Road, Donghu New Technology Development Zone, Wuhan

