AtaGenix Laboratories
AtaGenix Laboratories
Release time: 2025-08-22 View volume: 1143
Tumor Metabolic Reprogramming is a hallmark of cancer cells, characterized by the overactivation of glycolysis. Even under aerobic conditions, cancer cells rely on glycolysis to rapidly generate energy and metabolic intermediates. While RNA splicing abnormalities have been shown to influence tumor metabolism, the precise molecular mechanisms remain unclear. A study from Tongji Medical College, Huazhong University of Science and Technology, published in Cancer Research, focuses on the exon-skipping event of Ste20-like kinase (SLK). It reveals that the splice variant SLKv enhances glycolysis efficiency by directly acting on Enolase 1 (ENO1), offering new insights into the mechanisms of tumor metabolic reprogramming.
Splicing Abnormalities Globally Affect Tumor Metabolism
The study grouped samples from the ICGC cohort based on RNA splicing levels and found significant enrichment of metabolic pathways in the high-splicing group. Using the splicing inhibitor H3B-8800 in MHCC-97H cells, combined RNA-seq and metabolomics analyses showed that glycolysis and the TCA cycle were the most affected pathways. Data from TCGA for liver, breast, and colorectal cancers showed consistent trends. Gene expression analysis revealed significant changes in glycolysis/TCA-related genes, and Seahorse assays confirmed a decrease in ECAR and an increase in OCR, indicating that RNA splicing abnormalities are closely linked to tumor metabolic reprogramming.
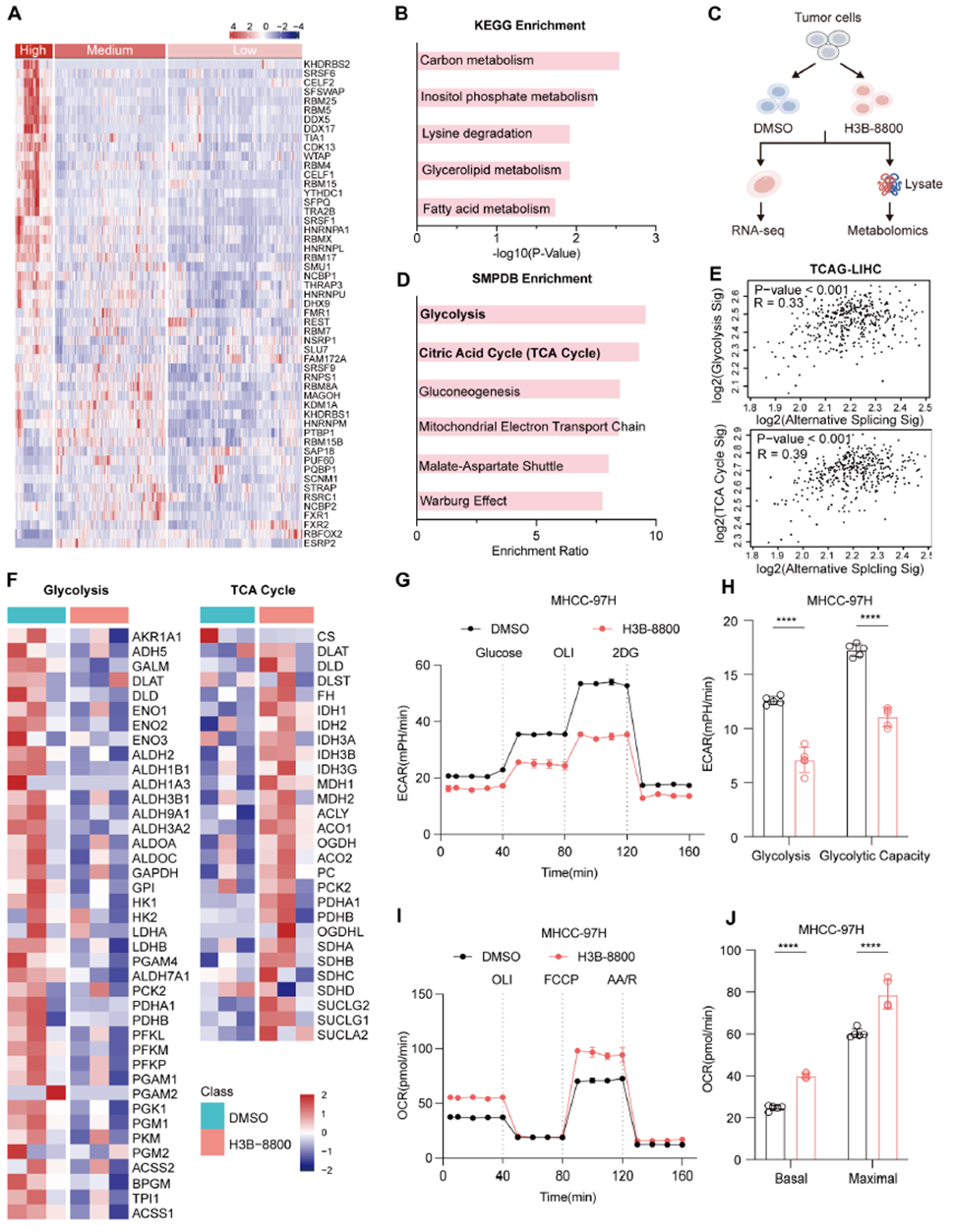
Fig. 1: RNA splicing abnormalities significantly impact glycolysis and the TCA cycle, driving tumor metabolic reprogramming
SLKv Is a Key Splicing Event Linked to Glycolysis
By comparing alternative splicing events across various tumor cells, the study identified that exon 13 skipping in the SLK gene produces a novel variant, SLKv, which is strongly associated with enhanced glycolysis. Splicing inhibition reduced SLKv expression, confirming its specificity as an aberrant splicing event. Clinical samples and patient imaging data showed that high SLKv expression correlated with increased glucose uptake, consistent with positive correlations to the glycolysis pathway in public databases. Functional experiments demonstrated that SLKv enhances cellular glycolysis and promotes tumor growth in animal models. In liver cancer patient samples, the SLKv/SLK ratio was significantly higher than in adjacent tissues, and high SLKv expression was associated with poorer prognosis, confirming that SLKv is a critical splice variant driving tumor glycolysis and progression.
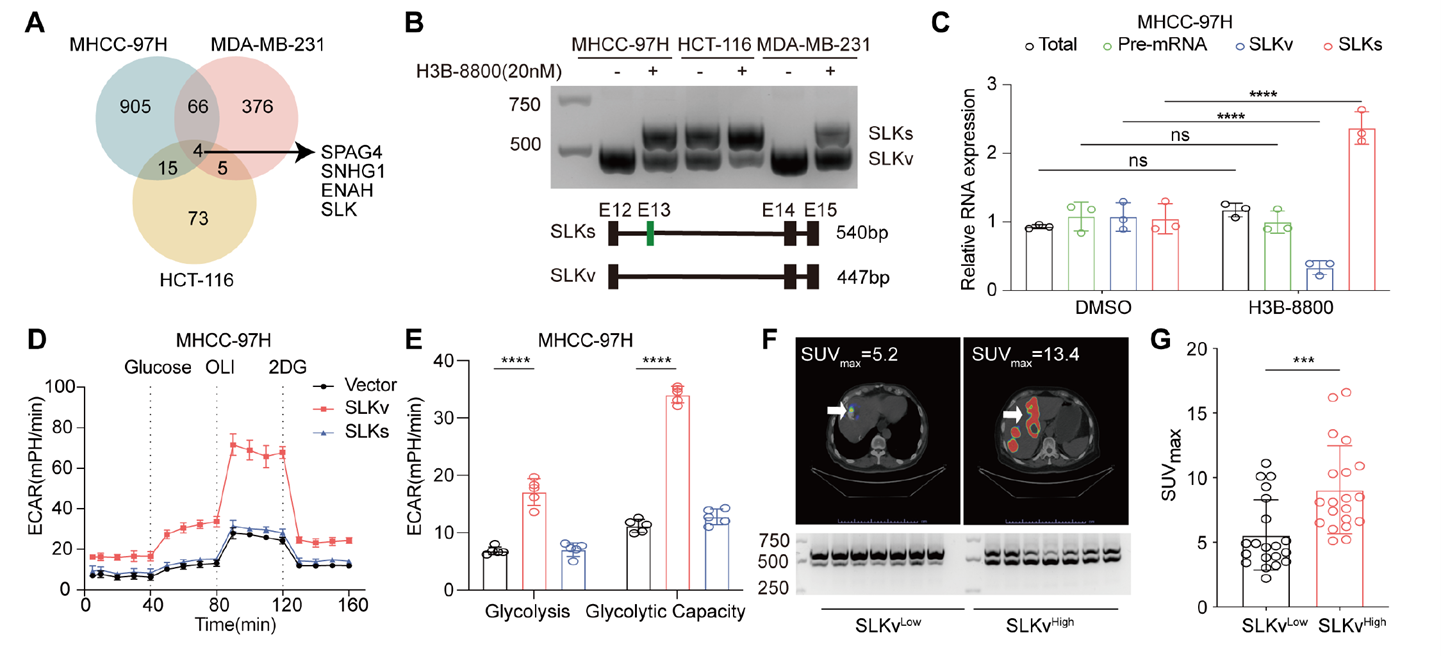
Fig. 2: The SLK exon 13 skipping variant SLKv is significantly associated with enhanced glycolysis and promotes tumor progression
SLKv Upregulates PEP, Driving Enhanced Glycolysis
Metabolic flux tracing and metabolomics revealed significantly elevated phosphoenolpyruvate (PEP) levels in SLKv-overexpressing cells. In xenograft experiments, this metabolic feature was accompanied by increased tumor volume and weight, while glycolysis inhibitors partially reversed this effect, indicating that PEP accumulation is a key metabolic step in SLKv-driven glycolysis and tumor growth.
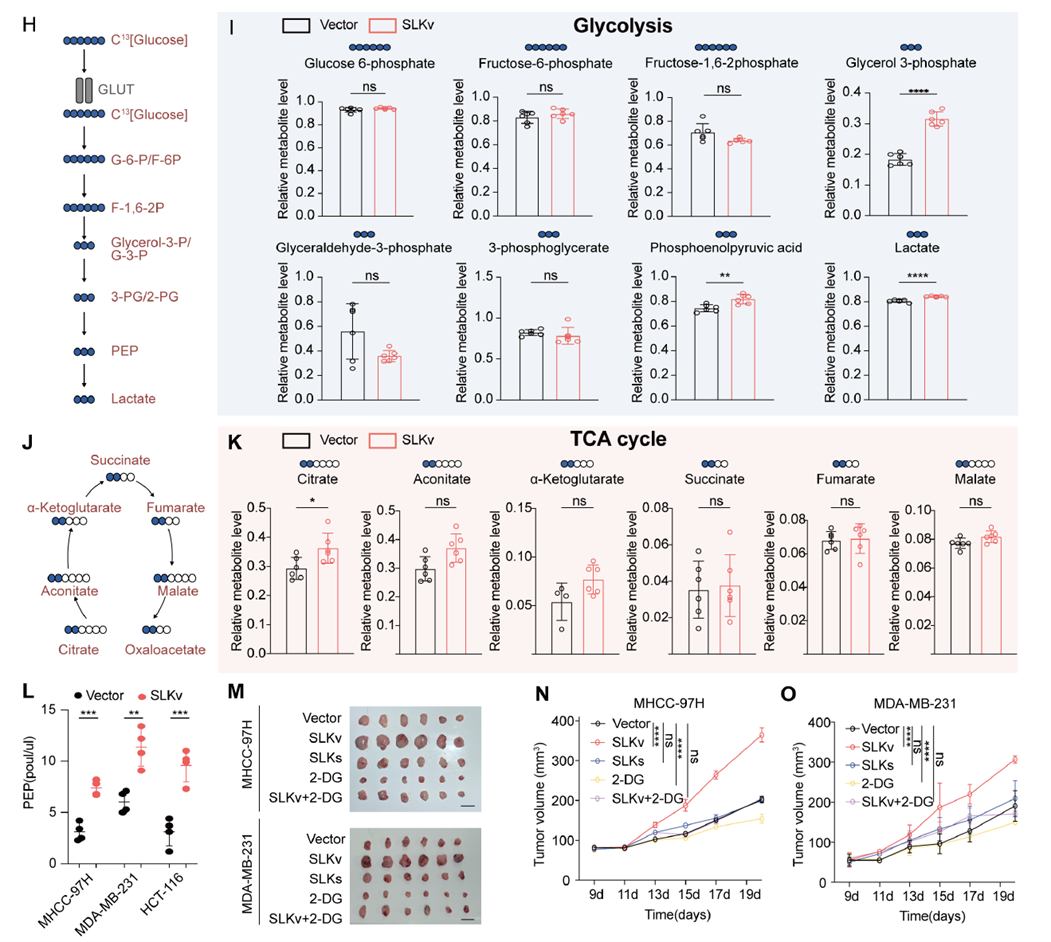
Fig. 3: SLKv drives PEP increase and enhances glycolysis
SLKv Acts Through ENO1, Not PEPCK
The study compared the roles of PEPCK and ENO1 and found that SLKv does not affect PEPCK but significantly enhances ENO1 enzyme activity. ENO1 inhibitors blocked SLKv-induced PEP increase and glycolysis, while PEPCK inhibitors had no effect. Binding assays confirmed that SLKv directly interacts with ENO1, dependent on ENO1’s C-terminal domain. ENO1 knockdown completely abolished SLKv’s promotion of glycolysis, indicating that SLKv exerts its key metabolic effects through ENO1, not PEPCK.
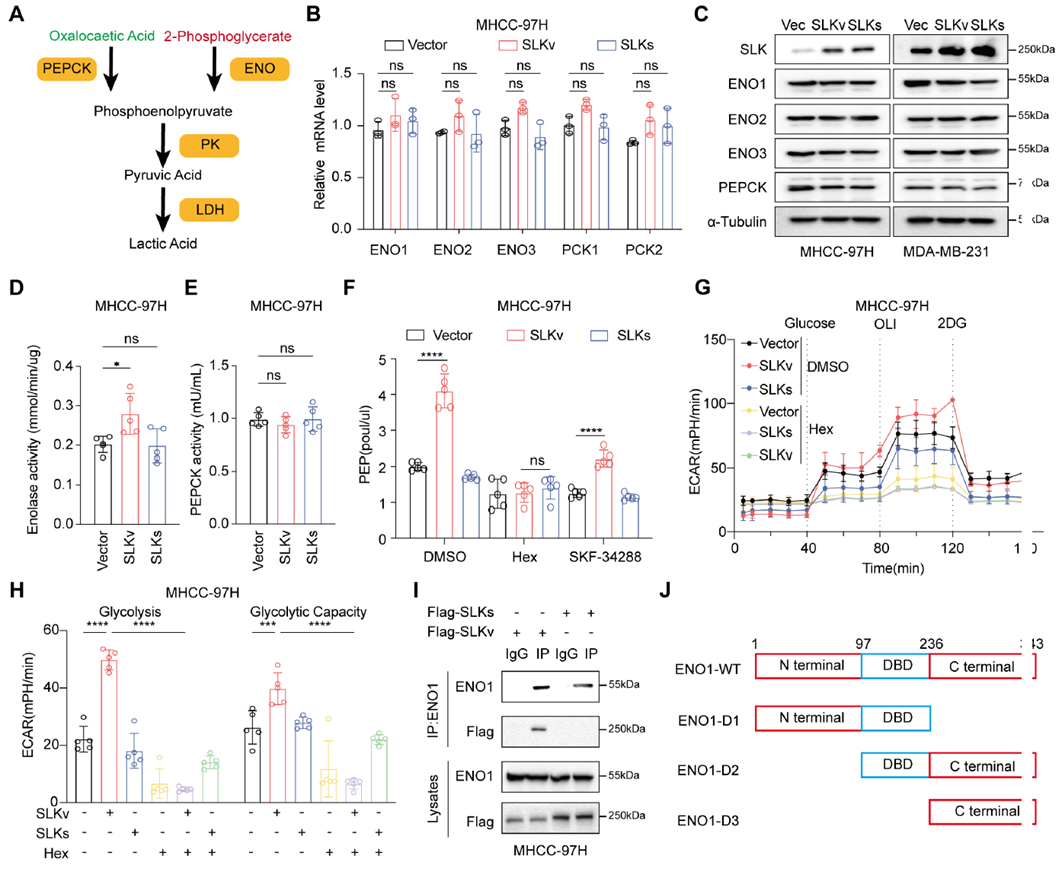
Fig. 4: SLKv-mediated ENO1-S2ph increases ENO1 activity to promote glycolysis and PEP synthesis
SLKv Directly Phosphorylates ENO1 at Ser2, Enhancing Its Activity
As a kinase, SLKv directly phosphorylates ENO1 at the Ser2 site, enhancing its activity. The standard SLK isoform lacks this effect, and kinase-inactive SLKv mutants lose functionality. Using a custom ENO1-S2 phosphorylation-specific antibody from AtaGenix, the study confirmed Ser2 modification and demonstrated its significant enhancement of ENO1 catalytic efficiency, driving PEP accumulation. This reveals a novel mechanism by which SLKv alters glycolysis flux through direct ENO1 modification.
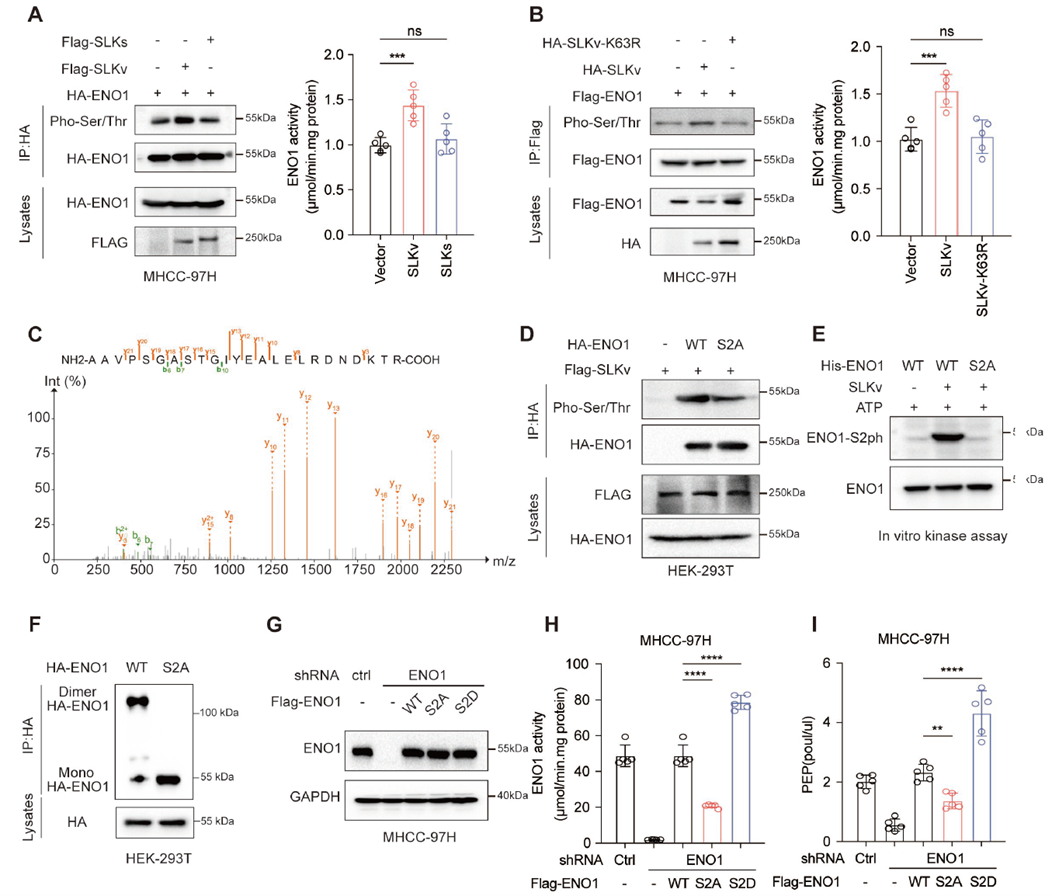
Fig. 5: Increased PEP promotes glycolysis in tumors by activating glycolytic enzymes
PEP Reversely Activates Key Glycolytic Enzymes, Forming a Positive Feedback Loop
The study found that PEP, beyond being a metabolic product, binds and activates key glycolytic enzymes such as HK2, PFKM, and PGAM1. CETSA and DARTS experiments further validated this role, showing that SLKv-driven PEP increase forms a self-reinforcing positive feedback loop, continuously enhancing glycolysis.
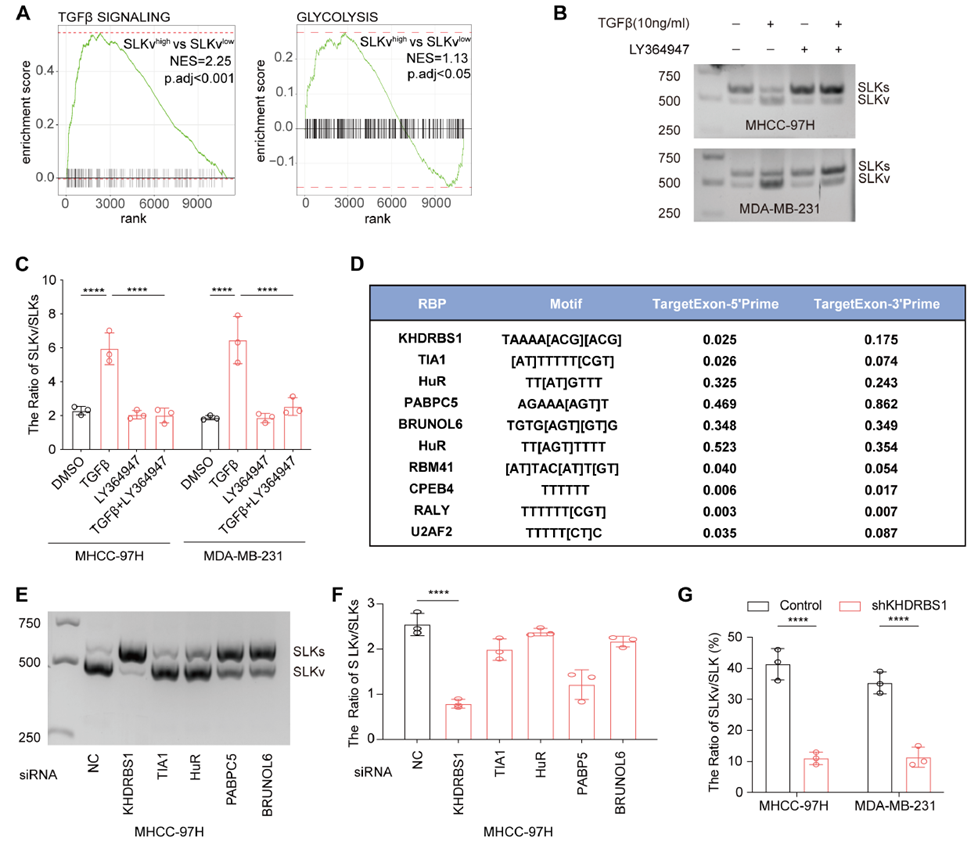
Fig. 6: The TGFβ/KHDRBS1 axis induces exon skipping in SLK
Upstream TGFβ–KHDRBS1 Signaling Drives SLKv Splicing
The study revealed that TGFβ signaling upregulates the splicing factor KHDRBS1, promoting SLK exon 13 skipping and increasing SLKv production. In animal experiments, antisense oligonucleotides (ASOs) targeting SLKv significantly reduced its expression, suppressed PEP accumulation and glycolysis, and decreased tumor growth, highlighting the therapeutic potential of targeting aberrant splicing events.
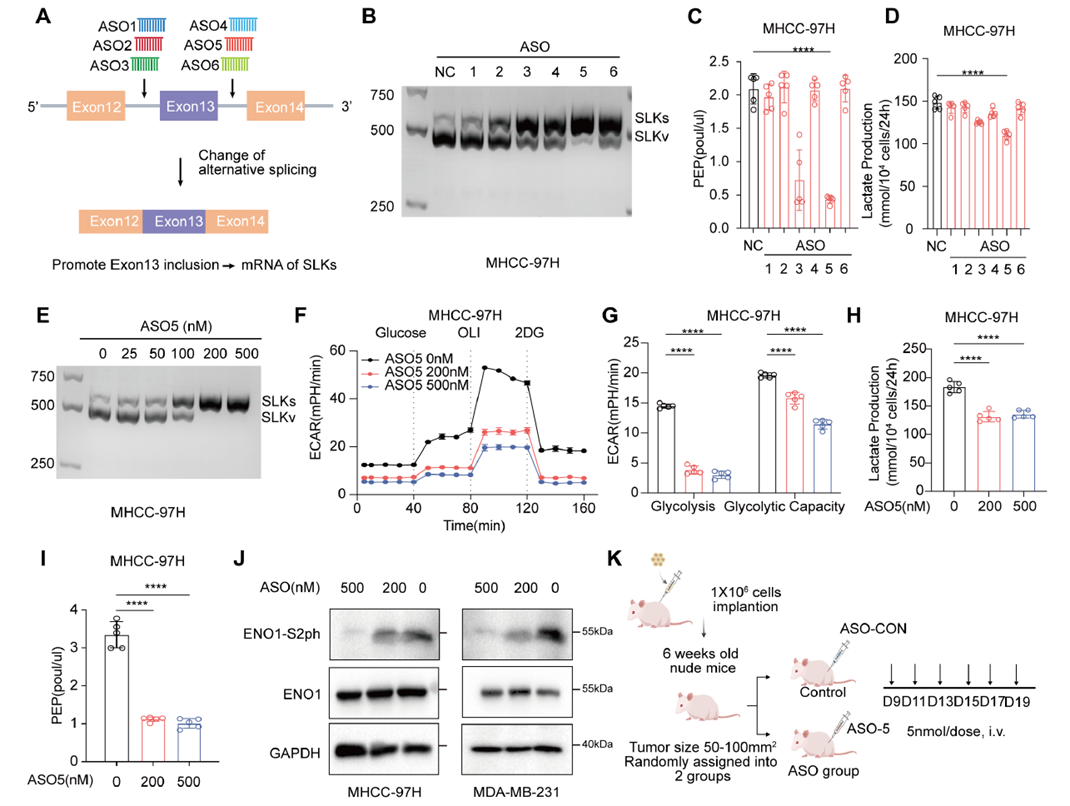
Fig. 7: ASOs targeting SLK pre-mRNA inhibit glycolysis and tumor growth
This study uncovers a novel mechanism by which ENO1, a rate-limiting enzyme in glycolysis, is activated by SLKv, and proposes PEP as a new “metabolic signaling molecule.” Combined with RNA splicing regulation, it provides a new perspective on the crosstalk between metabolism and signaling pathways, laying the foundation for combined anti-splicing/anti-metabolic therapeutic strategies. ASO therapy offers a feasible approach for tumor metabolic intervention.
AtaGenix provided critical ENO1-S2 (phospho-Ser2) phosphorylation-specific antibodies, ensuring high specificity and reliability in detecting phosphorylation modifications, supporting the validation of the SLKv-ENO1 regulatory mechanism. AtaGenix specializes in high-quality protein and antibody development, offering one-stop services from antibody discovery to large-scale production, accelerating scientific research.
To date, over 400 publications have cited AtaGenix’s one-stop protein and antibody development services. Looking ahead, AtaGenix will continue to support scientific innovation.
Contact Us
+86-27-6552-3339
info@atagenix.com
Building C, R & D Building, No. 666, Shendun 4th Road, Donghu New Technology Development Zone, Wuhan

