AtaGenix Laboratories
AtaGenix Laboratories
Release time: 2025-08-22 View volume: 1062
Perioperative Neurocognitive Disorders (PND) are common postoperative complications in patients over 60, including postoperative delirium (POD), delayed neurocognitive recovery (dNCR), and postoperative neurocognitive dysfunction (PNCD), with incidence rates up to 10%-40%. PND can lead to extended hospital stays, increased care costs, and long-term functional impairments, yet no FDA-approved prevention or treatment options exist.
Prior studies suggest the gut-brain axis may trigger central nervous system inflammation by activating microglia and astrocytes. The complement system's C3/C3aR pathway has recently been linked to neuroinflammation, but its precise role in elderly PND remains unclear. Additionally, platelet factor 4 (PF4), an aging-related "rejuvenation factor," has untapped potential in regulating neuroinflammation. In this study, researchers intervened in the C3 pathway using a selective C3 inhibitor, CR2-crry, custom-synthesized by AtaGenix, to systematically evaluate its role in postoperative neuroprotection. They also conducted recombinant PF4 supplementation experiments to uncover the interplay between complement and "rejuvenation factors" in PND.
In a randomized, double-blind clinical study, researchers intervened in 40 patients aged ≥60 undergoing oral cancer surgery, administering either short-acting cefazolin or long-acting ceftriaxone, with six-month postoperative follow-up. Results showed that the ceftriaxone group had significantly lower PNCD incidence at 30 and 180 days post-surgery compared to the cefazolin group.
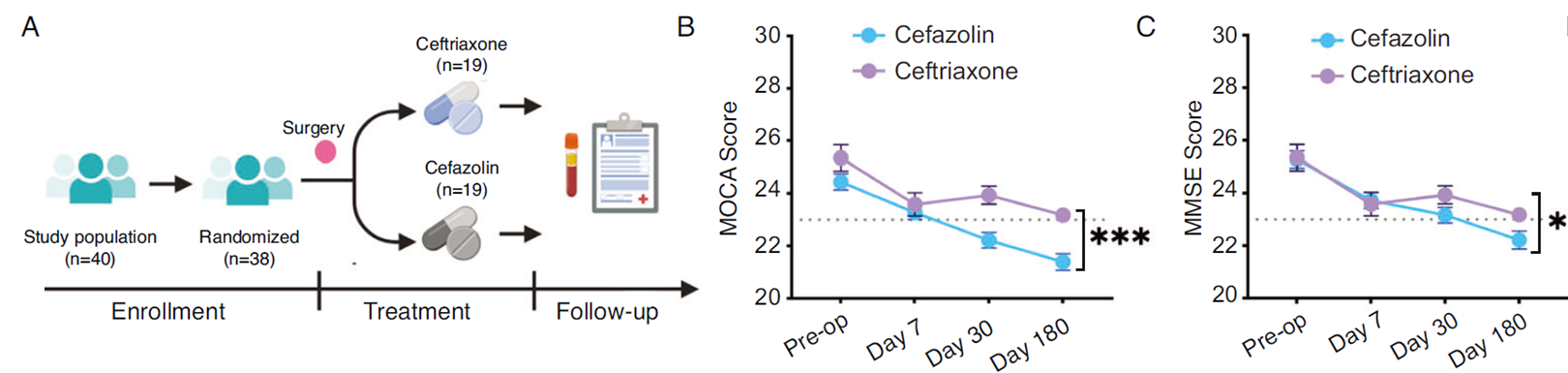
Fig. 1 Ceftriaxone significantly reduces postoperative PNCD incidence in elderly patients, improving cognitive function.
Corresponding to cognitive improvements, ceftriaxone markedly suppressed postoperative serum C3 elevation while maintaining higher platelet counts and PF4 levels. Spearman analysis revealed a negative correlation between C3 levels and cognitive scores, and a positive correlation between PF4 levels and cognitive scores. This suggests ceftriaxone may act through "C3 suppression + PF4 maintenance," reducing the risk of postoperative neurocognitive disorders in elderly patients and providing potential therapeutic targets. These findings laid the groundwork for subsequent animal studies to validate the causal mechanisms of the C3/C3aR pathway and PF4.
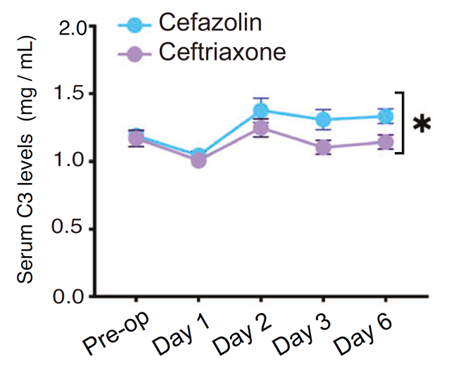
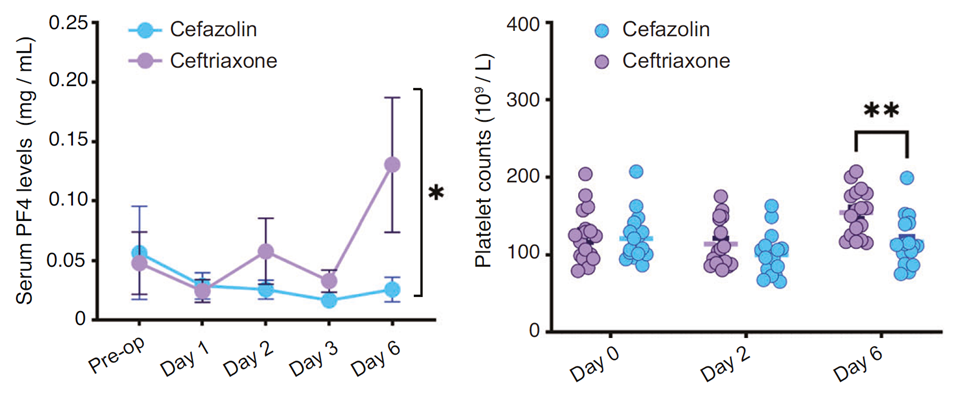
Fig. 2: Ceftriaxone suppresses C3 upregulation, maintains PF4 levels, and exerts cognitive protection.
Animal Models Validate C3/C3aR Pathway Activation of Glial Cells and Inflammatory Cascades
In an elderly mouse PND model, researchers observed the timing and cellular coordination of postoperative neuroinflammation: on day 1 post-surgery, astrocytes were activated first, significantly upregulating complement C3 expression; by day 3, microglial activation intensified, with peak expression of the characteristic receptor C3aR. Immunostaining showed C3 primarily localized in GFAP+ astrocytes, while C3aR was concentrated in Iba1+ microglia, indicating that the C3/C3aR pathway forms a "glial cell cascade activation," driving inflammation.
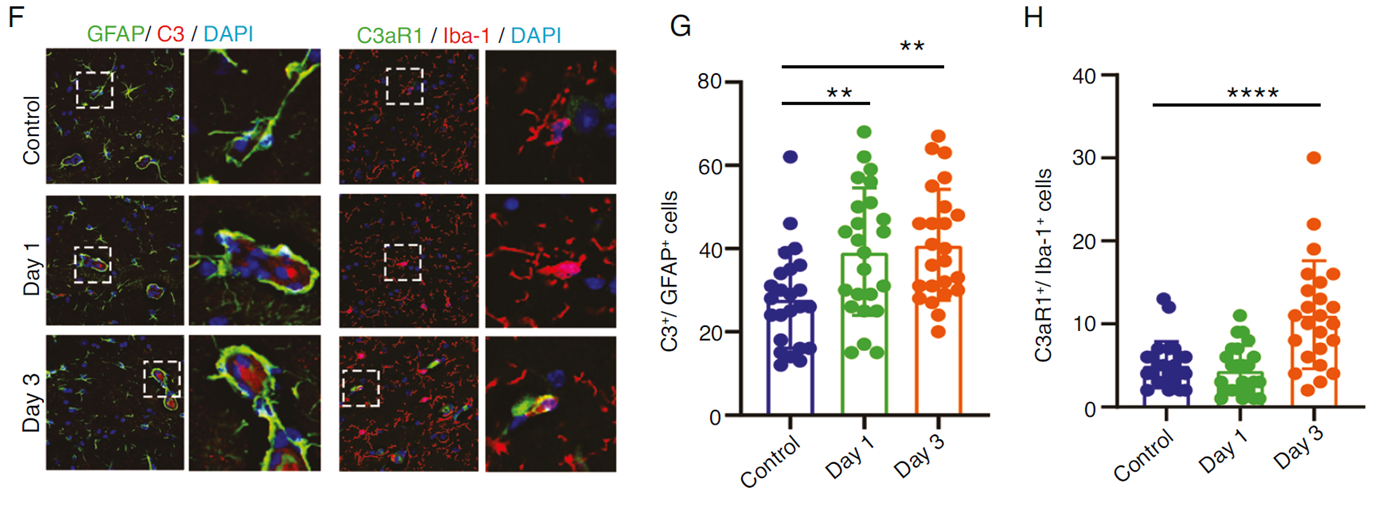
Fig. 3: Early postoperative C3/C3aR pathway mediates synergistic activation of astrocytes and microglia.
Concurrently, inflammation markers CD86 and iNOS were upregulated post-surgery, further confirming that early glial cell activation + C3/C3aR pathway activation = the core mechanism of PND-related neuroinflammation.
CR2-crry Intervention Blocks C3, Alleviating Inflammation and Cognitive Decline
After establishing the critical role of the C3/C3aR pathway in PND inflammation, researchers tested whether targeting this pathway could reverse pathological progression. Using the selective C3 inhibitor CR2-crry, administered preoperatively, they observed within one day: significant reductions in hippocampal inflammation markers—activated microglia (Iba1+), astrocytes (GFAP+), and complement C3 expression. Pro-inflammatory cytokines IL-6 and TNF-α levels decreased, and inflammatory effector proteins CD86 and iNOS were suppressed, indicating CR2-crry effectively interrupts the initiation of postoperative inflammatory pathways.
This inflammation suppression translated into "substantial functional benefits." In cognitive behavioral tests, CR2-crry significantly improved novel object recognition (NOR test) and contextual memory (FCT test), demonstrating clear cognitive protection. Further neuromorphological analysis confirmed these findings—Golgi-Cox staining showed CR2-crry treatment significantly enhanced neuronal dendritic branching complexity and synaptic spine density in postoperative mice, proving its neuroprotective effects.
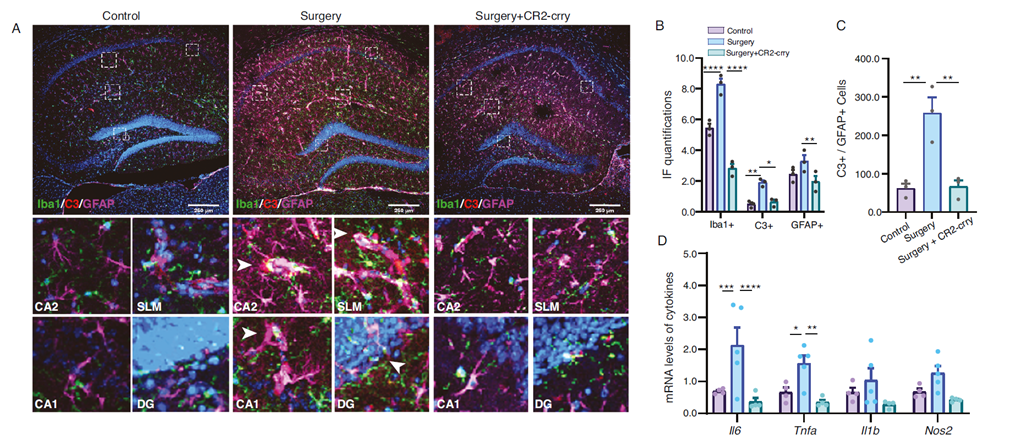
Fig. 4: CR2-crry significantly improves postoperative neuronal structure, enhancing dendritic branching and synaptic density.
Exploring PF4 in Reversing Cognitive Decline and Neuroinflammation
After elucidating the C3 pathway's dominant role in inflammation, researchers focused on its downstream key molecule—platelet factor 4 (PF4). They found that postoperative elderly mice exhibited significant platelet reduction and PF4 level decline, which was markedly alleviated in C3 knockout mice (C3⁻/⁻) (Fig. 5A–C), suggesting PF4 is regulated by the C3 pathway and is a critical link in C3-mediated neural injury.
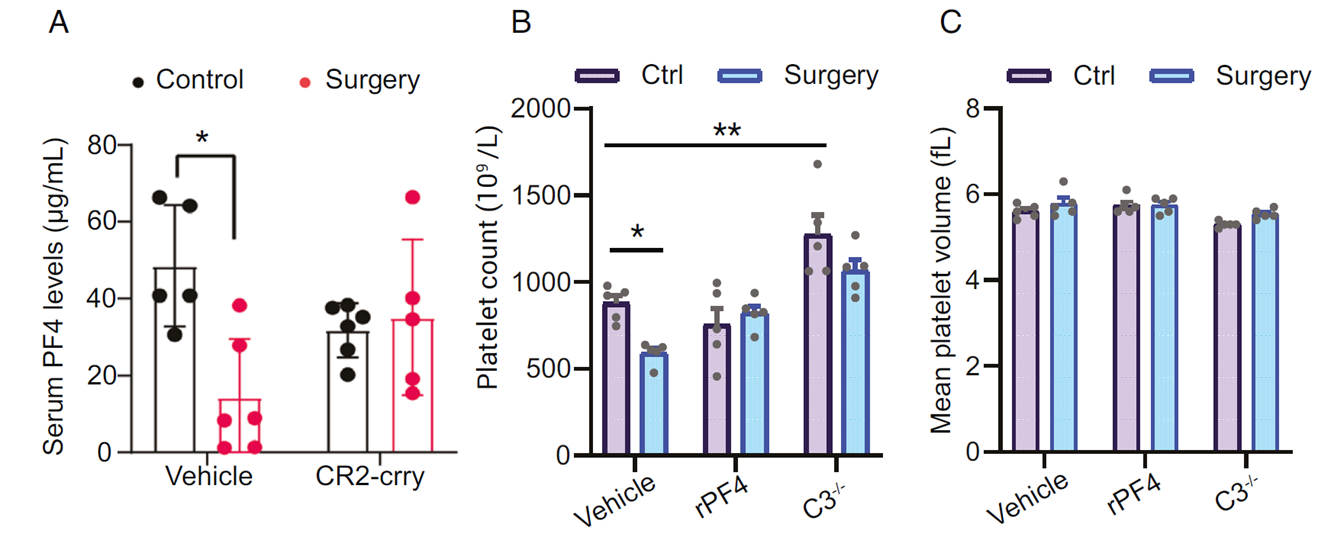
Fig. 5: Postoperative PF4 levels decline in elderly mice, significantly alleviated in C3 knockout mice.
To test PF4's ability to "counter" inflammation, researchers supplemented elderly mice with recombinant PF4 (rPF4). The results were encouraging: the PF4-treated group showed significant improvements in cognitive behavioral tests (OFT, NOR, and FCT), with comprehensive cognitive recovery. Moreover, neuronal dendritic structure and synaptic spine density were significantly restored, brain inflammatory cytokines IL-6 and TNF-α decreased (Fig. 5L), and glial cell activation was suppressed.
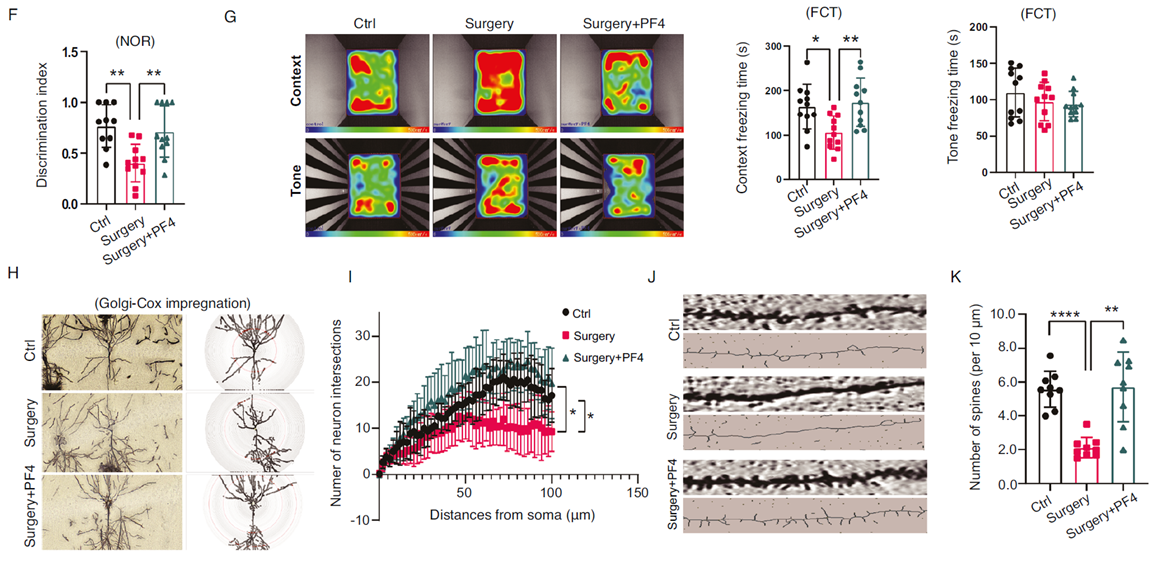
Fig. 6: PF4 improves and restores cognitive function.
In summary, this study systematically demonstrated that the complement C3/C3aR pathway not only promotes neuroinflammation and glial cell activation but also exacerbates cognitive impairment by affecting platelet function and PF4 secretion. As a blood-brain barrier-permeable factor, PF4 supplementation post-surgery effectively blocks these pathological cascades.
In this study, CR2-crry, custom-synthesized by AtaGenix, significantly inhibited complement activation, glial cell activation, and pro-inflammatory cytokine expression when administered intravenously preoperatively, ultimately protecting neural morphology and cognitive function. This suggests CR2-crry holds clinical translation potential for perioperative neuroprotection, providing precise molecular targets for future PND interventions.
AtaGenix's technology has helped researchers overcome numerous research bottlenecks, leading to significant discoveries. To date, over 400 publications have cited AtaGenix's one-stop protein and antibody development services. Looking forward, AtaGenix will continue to support scientific research, driving technological innovation and breakthroughs.
Contact Us
+86-27-6552-3339
info@atagenix.com
Building C, R & D Building, No. 666, Shendun 4th Road, Donghu New Technology Development Zone, Wuhan

