AtaGenix Laboratories
AtaGenix Laboratories
Release time: 2025-08-22 View volume: 703
Multiple sclerosis (MS) is an autoimmune disease characterized by chronic inflammation and demyelination of the central nervous system (CNS), with its relapsing nature posing significant challenges to patients’ quality of life and treatment. Although the role of B cells in MS pathology has gained increasing attention, the specific mechanisms by which they drive inflammation relapse in the meningeal microenvironment remain unclear. Recently, a research team from Westlake Laboratory of Life Sciences and Biomedicine published a significant study in Immunity, revealing that meningeal B cells drive MS neuroinflammatory relapse through antigen presentation and cognate interactions, offering new insights for precision therapy. Recent studies have found that the meninges, as a critical interface between the CNS and peripheral immunity, may contribute to disease relapse due to abnormal activation of local immune cells, particularly B cells. However, the precise mechanisms by which meningeal B cells break immune tolerance and trigger neuroinflammation remain to be fully elucidated.
The study focused on the role of meningeal B cells in the experimental autoimmune encephalomyelitis (EAE) model, employing single-cell RNA sequencing and immunofluorescence techniques to deeply analyze how B cells amplify inflammatory responses through interactions with T cells, providing a critical theoretical basis for targeted MS therapies.
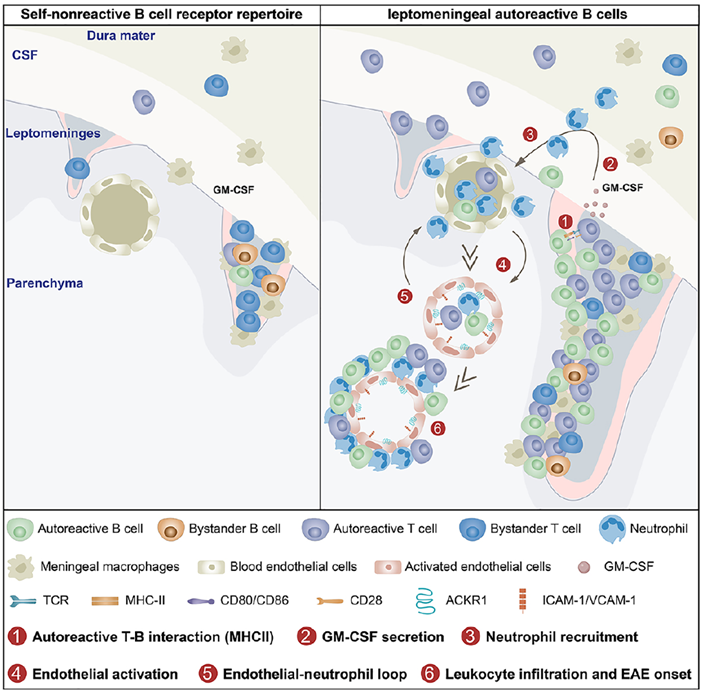
Fig. 1 Graphical abstract
Specific Activation of Autoreactive B Cells in the Pia Mater
The study first discovered that autoreactive B cells in the meninges are selectively activated under specific conditions, and this activation depends on CNS self-antigens. Through experiments involving intracranial injection of B cells in mice, researchers observed that these cells primarily accumulated in the pia mater, particularly in areas such as the subarachnoid cisterns of the pons and medulla, with minimal presence in peripheral tissues like the spinal cord or spleen. Autoreactive B cells from MOG-specific mice exhibited clear activation features in the pia mater, including downregulation of IgD, upregulation of activation markers, and high expression of antigen recognition-related genes. However, when experimental mice lacked the MOG antigen, these activation features disappeared, indicating that autoreactive B cell activation is driven by CNS self-antigens, confirming its direct association with the breakdown of central immune tolerance.
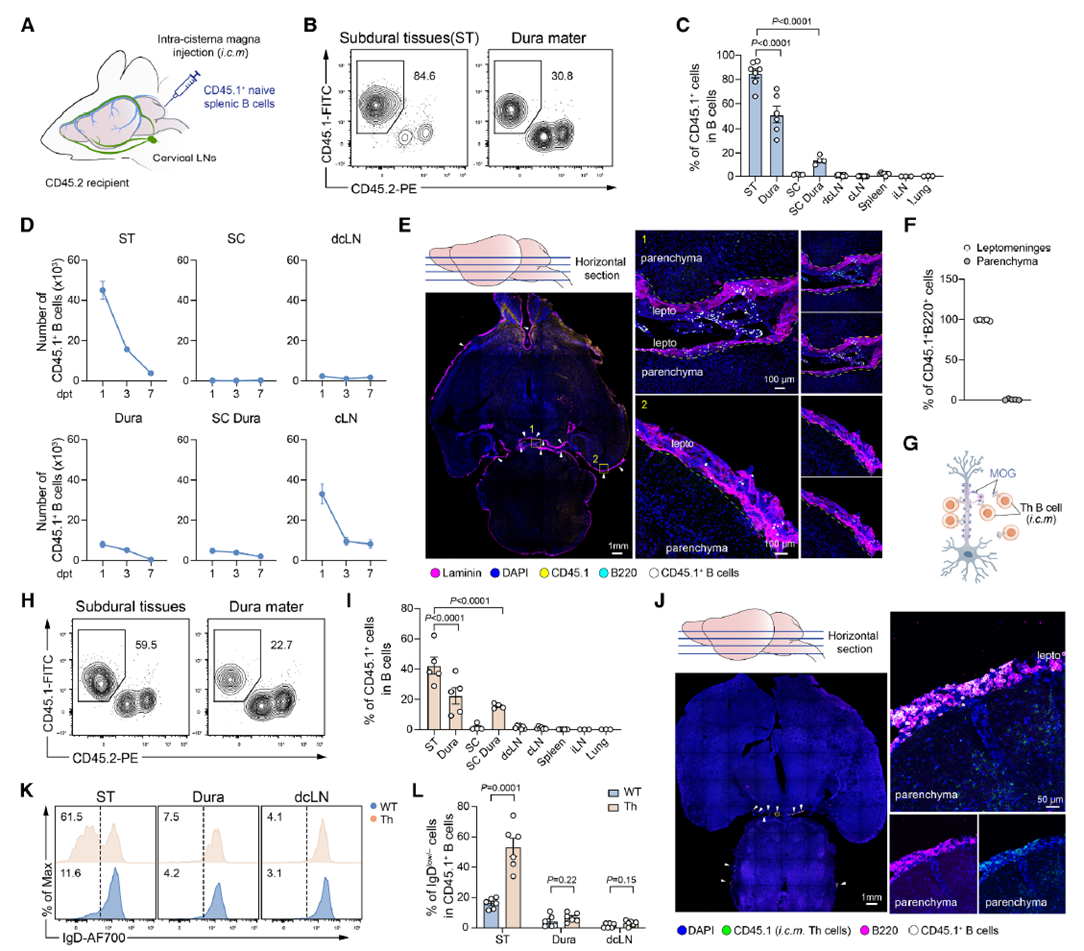
Fig. 2 Autoreactive B cells in the meninges promote T cell-mediated CNS inflammation through antigen presentation
B Cells Accelerate T Cell-Mediated Neuroinflammation via Antigen Presentation
Further studies revealed that autoreactive B cells in the meninges exacerbate CNS inflammatory responses through specific interactions with pathogenic T cells. In the EAE model, mice receiving intracranial injections of autoreactive B cells showed significantly earlier disease onset and more severe symptoms, whereas intravenous injections of the same B cells had no similar effect, indicating that this pathogenic role is unique to local meningeal B cells. By excising cervical lymph nodes or blocking meningeal lymphatic drainage, researchers ruled out the influence of peripheral immune organs, confirming that meningeal autoreactive B cells can independently drive inflammation. Moreover, when autoreactive B cells lacked MHC-II molecules, their ability to promote inflammation was completely lost, and T-B cell aggregates in the brain decreased, suggesting that these B cells activate pathogenic T cells through MHC-II-mediated antigen presentation, thereby accelerating disease progression.
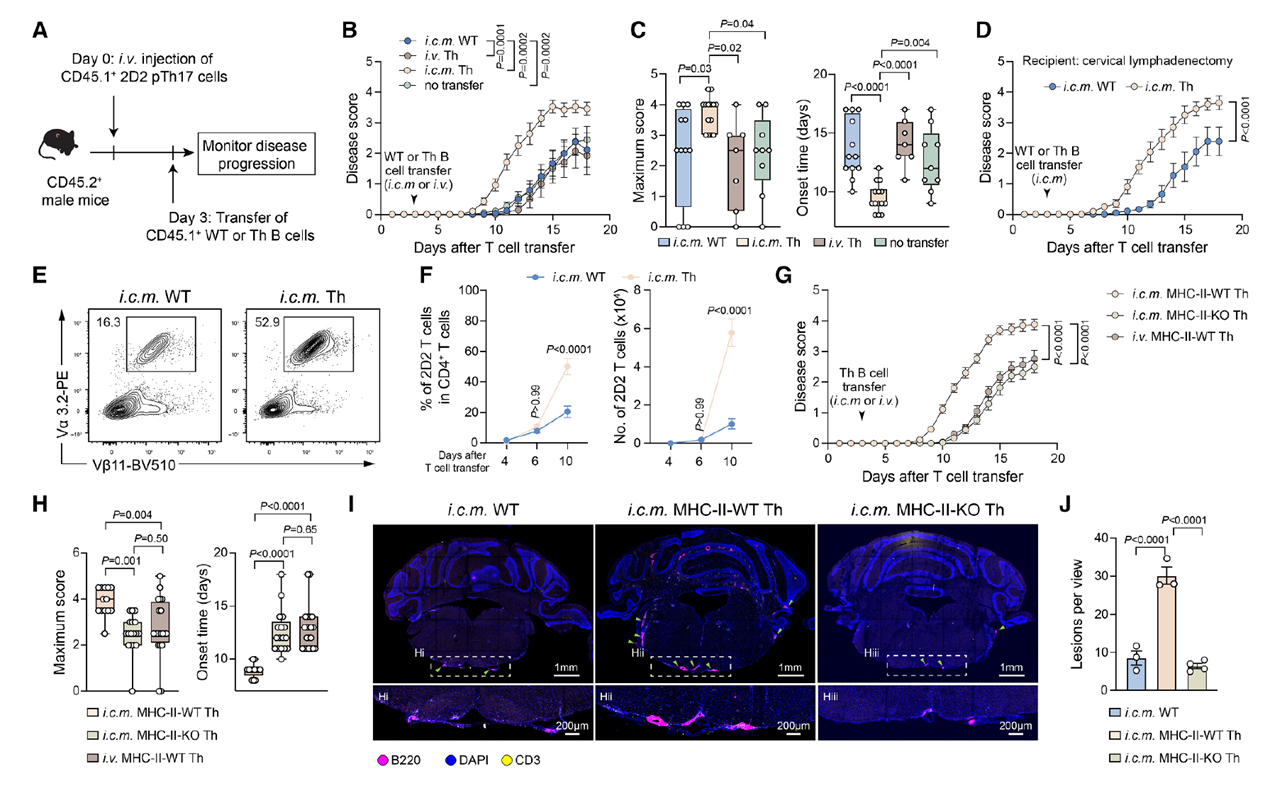
Fig. 3 Autoreactive B cells in the meninges promote T cell-mediated CNS inflammation through antigen presentation
B-T Cell Interactions Initiate an Inflammatory Amplification Loop
The study further explored the inflammatory amplification mechanism triggered by interactions between meningeal autoreactive B cells and T cells, which forms a “T-B cell-neutrophil-endothelial cell” inflammatory loop, driving the sustained progression of neuroinflammation. Experiments showed that after autoreactive B cells interacted with T cells, T cells produced large amounts of GM-CSF, a cytokine that further recruited neutrophils to the meningeal region. Once recruited, neutrophils activated brain vascular endothelial cells, leading to increased expression of adhesion molecules such as VCAM-1 and ACKR1, which facilitated further leukocyte infiltration into the CNS, creating a continuously amplifying inflammatory cycle. Experiments using GM-CSF-deficient T cells or neutrophil depletion confirmed that GM-CSF is critical for neutrophil recruitment, while neutrophils are essential for endothelial cell activation in this loop.
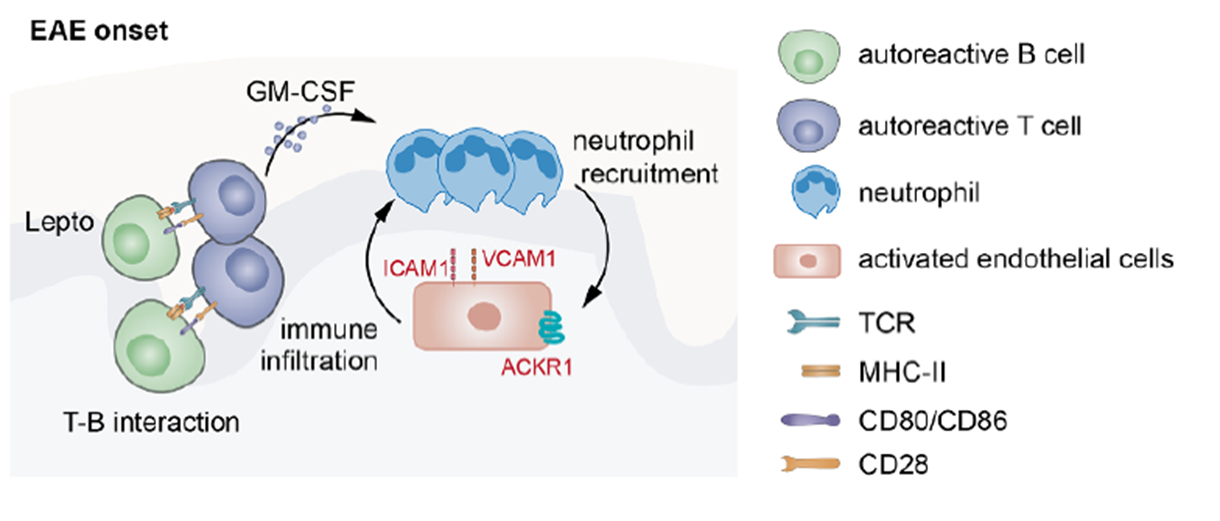
Fig. 4. GM-CSF-dependent neutrophil recruitment and endothelial cell activation in the pro-inflammatory loop
Targeted Depletion of Local Brain B Cells Alleviates EAE Relapse
Finally, the study demonstrated that targeted depletion of local autoreactive B cells in the brain effectively alleviates neuroinflammatory relapse, offering new directions for treating related diseases. In a relapsing-remitting EAE model, intracranial injection of low-dose anti-CD20 antibodies selectively depleted B cells in the brain without affecting those in peripheral tissues. The results showed that this treatment significantly reduced relapse symptoms in mice, with decreased T cell proliferation, reduced production of pro-inflammatory cytokines, and a notable decline in activated endothelial cell numbers. Single-cell sequencing analysis revealed significant clonal expansion of B cells in the brain during inflammatory relapse, with these clonal populations persisting during remission, further supporting the role of local brain B cells as drivers of relapse and providing a theoretical basis for targeted therapies.
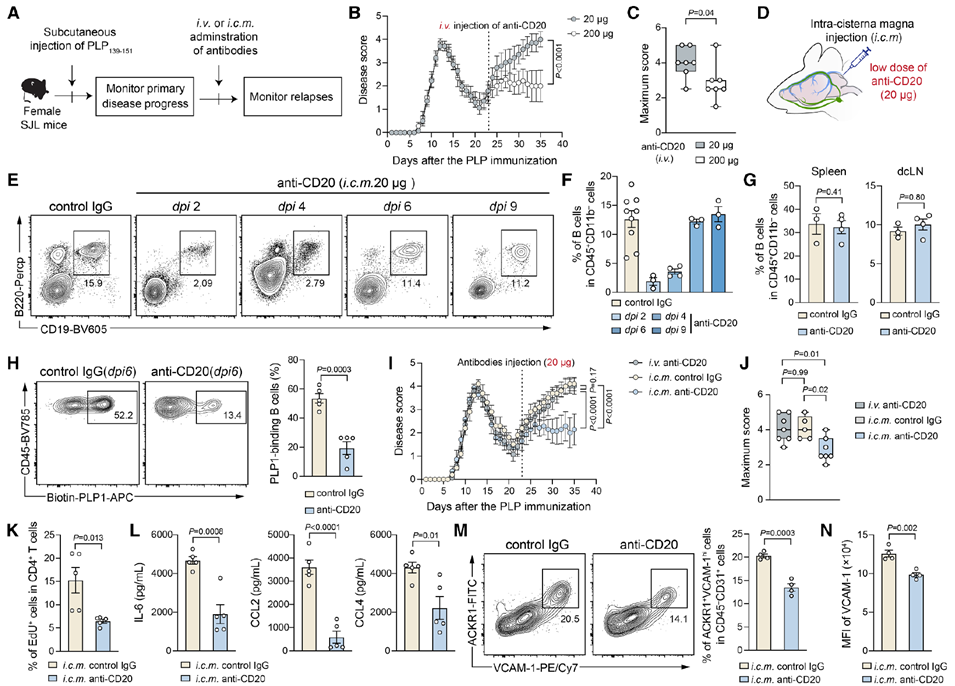
Fig. 5. Targeted depletion of local brain B cells alleviates EAE relapse
This study is the first to systematically reveal the molecular mechanisms by which meningeal B cells drive MS inflammatory relapse through cognate interactions, providing critical evidence for B cell-targeted therapies and filling a key gap in MS pathology research. Additionally, through targeted B cell depletion experiments, the study validated the feasibility of meningeal B cells as therapeutic targets, offering theoretical support for developing novel anti-B cell therapies (such as anti-CD20 or CD40 inhibitors).
AtaGenix Laboratories provided the research team with high-purity, highly bioactive recombinant mouse MOG protein, used in EAE model construction and antigen presentation experiments. The MOG protein underwent stringent quality control, ensuring stability and reproducibility of antigen induction in experiments, providing reliable support for validating key mechanisms of B cell-T cell interactions.
AtaGenix’s technology has helped researchers overcome numerous research bottlenecks, leading to a series of significant discoveries. To date, over 400 publications have cited AtaGenix’s one-stop protein and antibody development technical services. Looking ahead, AtaGenix will continue to fully support scientific research, driving technological innovation and breakthroughs.
Contact Us
+86-27-6552-3339
info@atagenix.com
Building C, R & D Building, No. 666, Shendun 4th Road, Donghu New Technology Development Zone, Wuhan

