AtaGenix Laboratories
AtaGenix Laboratories
Release time: 2025-07-24 View volume: 1615
A practical guide to protein tag selection for recombinant expression and purification. Covers His, GST, Flag, MBP, HA, Strep-tag II, Twin-Strep, and SUMO — including advantages, limitations, and combination strategies across prokaryotic, mammalian, and insect cell expression systems.
In recombinant protein work, “tagging” is standard practice for enabling expression, detection, and purification. But tag selection is far from one-size-fits-all — a poor choice can reduce expression levels, lower purification efficiency, or cause abnormal protein structure and loss of function. This article summarizes lessons learned and practical combinations to guide your experimental design.
The His tag (typically 6×His, ~0.8 kDa) is the most widely used affinity purification tag, offering simple Ni²+/Co²+ affinity chromatography with low cost and minimal impact on target protein structure. However, non-specific binding can occur, requiring optimized imidazole elution gradients. Detection sensitivity is relatively low for WB, so combining with other tags (Flag, HA) is common.
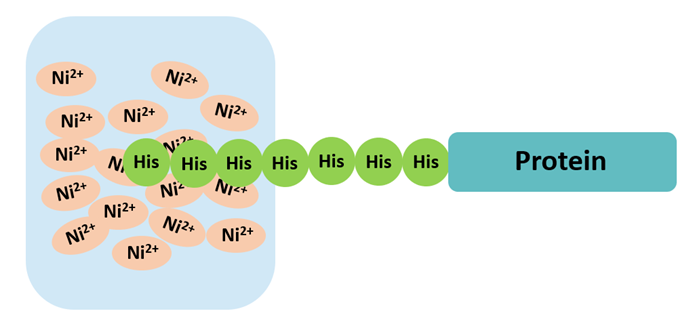
Fig 1. His-tag purification principle via Ni²+ affinity chromatography.
GST (211 aa, 26 kDa) enhances solubility of recombinant proteins and binds glutathione (GSH) for affinity purification. Commonly used in GST pull-down assays for protein–protein interaction studies. Its large size may affect native conformation — remove with specific proteases (PreScission, thrombin) after purification for structural/functional studies.
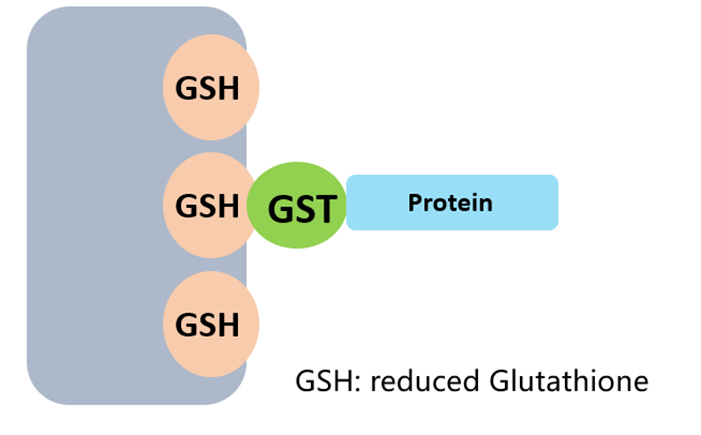
Fig 2. GST tag purification via glutathione affinity chromatography.
Flag is a short hydrophilic peptide with excellent antibody recognition, ideal for WB, IP, and IF detection with high specificity and low background, particularly in mammalian expression systems. Not suitable for standalone affinity purification (anti-Flag columns are costly) — best paired with His for a detection + purification dual-tag strategy.
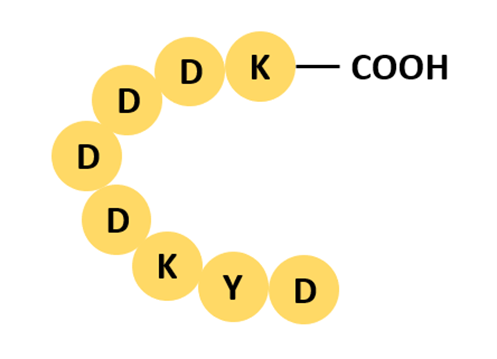
Fig 3. Flag tag structure (DYKDDDDK).
MBP (~42 kDa) is highly effective at increasing soluble expression of inclusion body-prone proteins in prokaryotic systems. Purification uses amylose affinity columns. Like GST, its large size may interfere with target protein function — enzymatic cleavage recommended for downstream structural or functional studies.
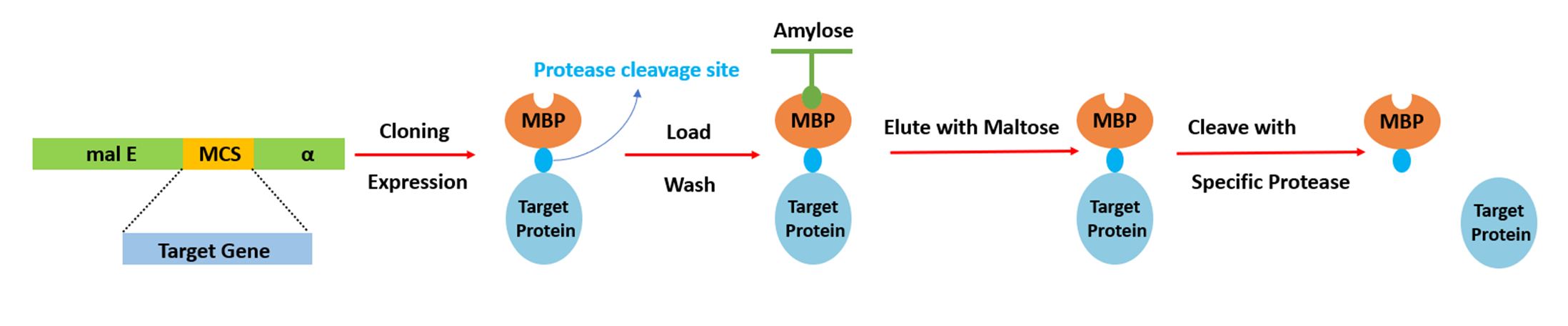
Fig 4. MBP tag purification via amylose affinity chromatography.
The HA tag (9 aa, derived from influenza hemagglutinin) binds specific anti-HA antibodies for WB, IP, IF, and intracellular localization. Widely used in dual-tag systems (e.g., HA + Flag or HA + His) for co-expression and co-IP experiments.
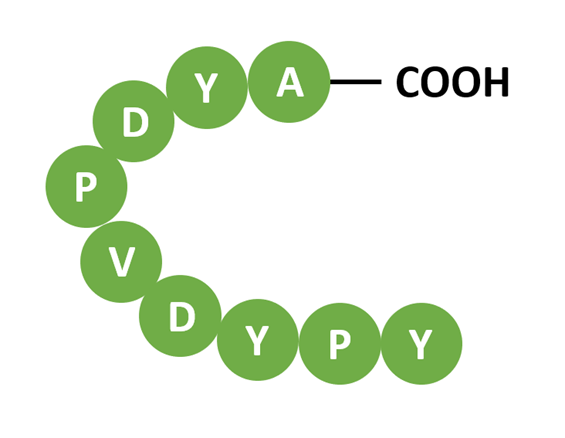
Fig 5. HA tag structure (YPYDVPDYA).
Strep-tag II (8 aa, WSHPQFEK) binds Strep-Tactin resin for gentle affinity purification that preserves protein activity — ideal for structural and functional studies. Twin-Strep-tag (two Strep-tag II units linked by flexible peptide) offers >10× higher affinity, enabling efficient purification of low-abundance proteins from complex samples. Slightly higher cost and specialized resin required.
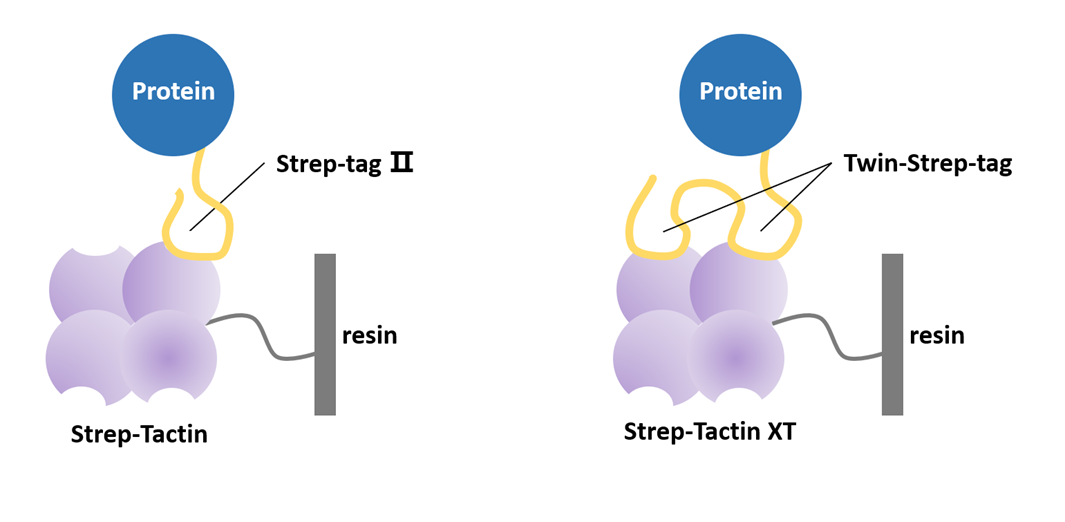
Fig 6. Strep-tag purification via Strep-Tactin affinity chromatography.
SUMO (~12 kDa) enhances both solubility and stability of fusion proteins. SUMO-specific proteases (Ulp1/SENP) precisely remove the tag to restore the native N-terminus, making it particularly valuable for sensitive proteins in functional and structural biology studies.

Fig 7. SUMO tag fusion protein structure with cleavable linker.
His
Small, cheap, routine purification
GST
Solubility + pull-down assays
Flag
High-specificity detection (WB/IP/IF)
MBP
Inclusion body rescue in E. coli
HA
Detection + dual-tag systems
Strep / Twin-Strep
Gentle purification, preserves activity
SUMO
Solubility + cleavable, native N-term
Need help with tag selection, expression strategy, or purification optimization? AtaGenix offers multi-system protein expression services with customizable His, GST, Flag, MBP, Strep, and SUMO tags across prokaryotic, mammalian, and insect cell platforms.
Talk to Technical SupportResponse within 24 hours
Contact Us
+86-27-6552-3339
info@atagenix.com
Building C, R & D Building, No. 666, Shendun 4th Road, Donghu New Technology Development Zone, Wuhan

