AtaGenix Laboratories
AtaGenix Laboratories
Release time: 2024-10-21 View volume: 748
Overview — AtaGenix's proprietary XtenCHO™ high-density transient expression system delivers therapeutic antibody yields of 500–1,000 mg/L across diverse molecules, with top performers reaching ~1,500 mg/L. In 15-day culture runs, CHO cells maintained near-100% viability — providing a stable, scalable platform that shortens development timelines and reduces production risk for antibody drug candidates.
Transient expression in CHO cells is the fastest route from antibody sequence to purified protein — but conventional systems often hit a ceiling. Cell viability drops after day 5–7, limiting culture duration and final titer. Yields for "average" antibodies hover around 100–300 mg/L, and difficult molecules (bispecifics, heavily glycosylated variants, non-standard Fc backbones) may fall below 50 mg/L. For programs that need milligram-to-gram quantities for preclinical studies, this means either multiple transfection batches (inconsistent) or early commitment to stable cell line development (slow and expensive).
XtenCHO™ was engineered to close this gap: a genetically optimized CHO host with proprietary media formulation that sustains high cell density and viability for 15 days, pushing transient yields into the range typically associated with stable pools — without the 8–12 week timeline of cell line development.
In head-to-head comparisons across multiple CHO lines carrying different antibody constructs, XtenCHO™ demonstrated consistent growth kinetics and exceptional viability retention:
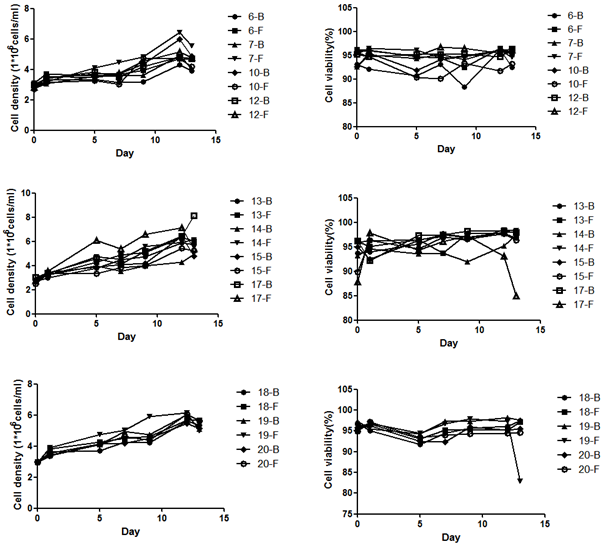
Figure 1. Cell density and viability across multiple CHO lines over 15 days. Most lines expanded steadily to high density while maintaining near-100% viability through day 12–13. Minor viability decline near day 15 in select lines is typical of extended culture and manageable through optimized feeding strategies.
Key observations:
To demonstrate platform versatility, AtaGenix benchmarked XtenCHO™ across 11 different therapeutic antibody molecules spanning multiple IgG subclasses and engineered formats:

Figure 2. Antibody yields across 11 therapeutic molecules expressed in XtenCHO™. The majority of molecules achieved 500–1,000 mg/L, with the top performer reaching ~1,500 mg/L. A small number of outliers below 100 mg/L reflect sequence-specific challenges (e.g., secretion bottlenecks, glycan complexity) that can typically be addressed through leader peptide optimization or process adjustments.
What the yield data tells us:
| Parameter | XtenCHO™ | Standard CHO Transient |
|---|---|---|
| Typical Yield | 500–1,000 mg/L | 100–300 mg/L |
| Top Titer | ~1,500 mg/L | ~500 mg/L |
| Culture Duration | 15 days (high viability) | 5–7 days |
| Viability at Harvest | >90% | 60–80% |
| Turnaround | ~3 weeks (gene to protein) | ~2–3 weeks |
| Best For | Preclinical material, lead optimization, multi-molecule panels | Early screening, small-scale feasibility |
Why This Matters
In antibody drug development, the gap between "enough protein to characterize" and "enough protein for preclinical studies" is where projects stall. Standard transient expression delivers the former; stable cell lines deliver the latter — but take 12+ weeks. XtenCHO™ fills this gap by delivering stable-line-level yields in transient timelines. For programs evaluating multiple candidates in parallel, this means generating gram-scale material for 5–10 molecules simultaneously, selecting the best performers, and then investing in stable cell line development only for the winners. It's a smarter allocation of time and budget at the stage where both are most constrained.
Yield data based on internal benchmarking across 11 therapeutic antibody molecules. Results may vary depending on antibody sequence, format, and process conditions. XtenCHO™ is a proprietary platform of AtaGenix Laboratories.
Need gram-scale antibody material in transient timelines? AtaGenix's XtenCHO™ platform delivers 500–1,500 mg/L yields with process development and optimization support included.
Discuss Your Antibody Expression ProjectResponse within 24 hours
Contact Us
+86-27-6552-3339
info@atagenix.com
Building C, R & D Building, No. 666, Shendun 4th Road, Donghu New Technology Development Zone, Wuhan

