AtaGenix Laboratories
AtaGenix Laboratories
Release time: 2025-10-14 View volume: 691
Project Snapshot — A neuroscience team studying truncated tau pathology in Alzheimer’s disease needed a highly specific monoclonal antibody to distinguish truncated tau N1-368 from full-length tau in a novel tet-on transgenic mouse model (hTau368). AtaGenix delivered a custom anti-tauN368 mAb via hybridoma technology with ELISA titer >1:64,000 and >95% purity, validated for WB, IF, and IHC — enabling a publication in Translational Neurodegeneration (2023).
Alzheimer’s disease (AD) is the most common neurodegenerative disorder in the elderly, driven by abnormal accumulation of hyperphosphorylated tau protein that forms neurofibrillary tangles (NFTs) and causes cognitive decline. Truncated tau fragments — particularly N1-368 — are significantly elevated in AD patient brains and aged mice, promoting tau pathology progression. To model this mechanism specifically, researchers developed a novel tet-on transgenic mouse model (hTau368) that inducibly expresses truncated human tau N1-368, producing hippocampal tau accumulation, phosphorylation, neuronal loss, and cognitive impairment. The related research was published in Translational Neurodegeneration (2023, DOI: 10.1186/s40035-023-00379-5).
The research team aimed to establish and characterize an accessible AD animal model. They required a monoclonal antibody that could: (1) distinguish truncated tau N1-368 from full-length tau with high specificity and zero cross-reactivity; (2) work reliably across multiple platforms — Western Blot, immunofluorescence (IF), and immunohistochemistry (IHC); (3) detect tau accumulation in specific hippocampal subregions (CA1, DG) with sensitivity sufficient for quantitative analysis; and (4) provide stable, reproducible performance to validate a doxycycline-controlled reversible expression system across multiple experimental timepoints.
Developing an antibody against the tau truncation site presented specific difficulties:
AtaGenix designed a targeted workflow to address each challenge:
With AtaGenix’s anti-tauN368 antibody, the research team successfully validated doxycycline-induced reversible tau accumulation and phosphorylation in the hTau368 hippocampus. The antibody confirmed that truncated tau triggers glial activation, neuronal loss, and synaptic degeneration, directly linking these events to cognitive impairment measured by Morris water maze behavioral testing. Electron microscopy further corroborated synaptic damage. These findings established hTau368 as an accessible, controllable AD model for tau-targeted drug development — a significant advance beyond traditional Aβ-focused approaches. For the client, the project delivered publication-quality data and a validated detection tool for ongoing translational research in tau-targeted therapeutics.

Figure 1. Characterization of the hTau368 transgenic mouse model. Doxycycline-induced expression of truncated human tau N1-368 in the hippocampus, detected and validated using AtaGenix custom anti-tauN368 monoclonal antibody.
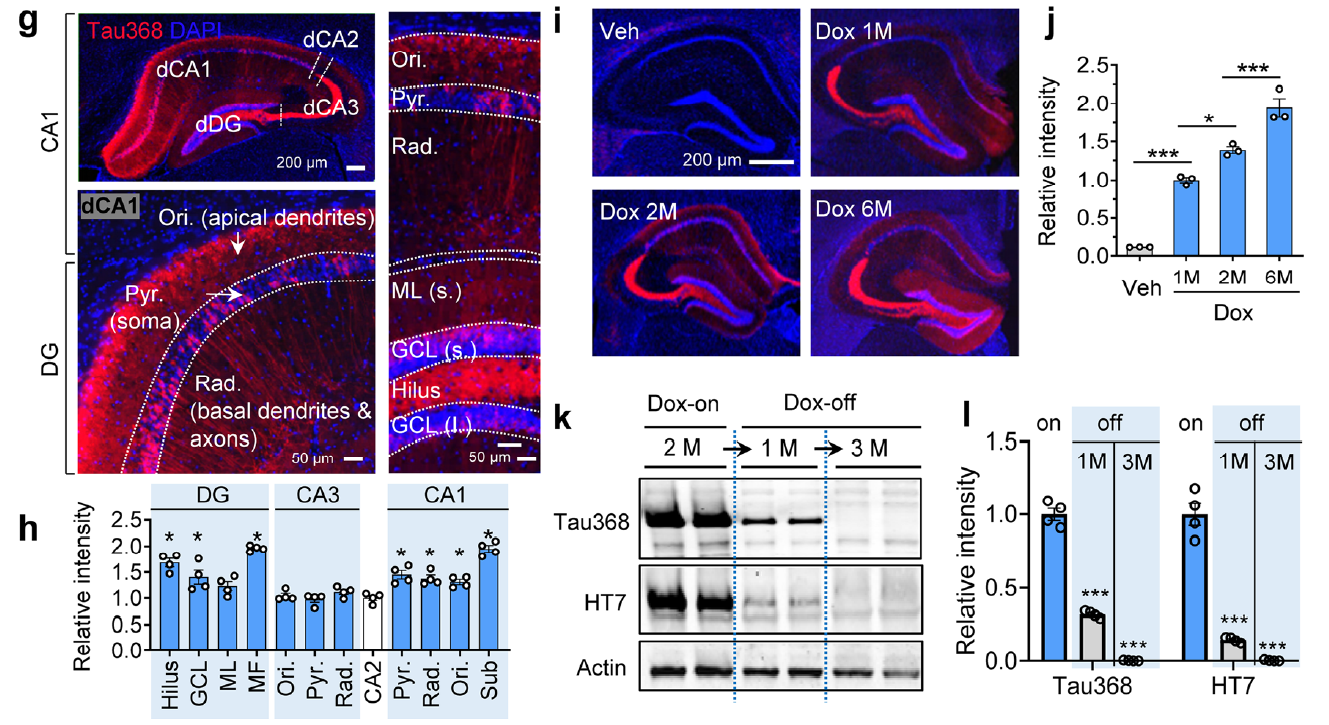
Figure 2. Tau accumulation and phosphorylation in hippocampal CA1 and DG subregions. Anti-tauN368 WB and IF analysis confirmed region-specific truncated tau expression, phosphorylation, and reversibility upon doxycycline withdrawal.
About AtaGenix
AtaGenix specializes in one-stop custom antibody and protein solutions, from antigen design and peptide synthesis through hybridoma development, recombinant expression, and multi-platform validation. Learn more about our services at www.atagenix.com.
This case study is based on a published research collaboration. Results may vary depending on target antigen, antibody format, and experimental conditions. All proprietary client information is subject to NDA. Reference: Zhang Y, et al. Translational Neurodegeneration. 2023. DOI: 10.1186/s40035-023-00379-5
Need a custom neo-epitope antibody for truncation sites, cleavage products, or disease-specific protein forms? AtaGenix delivers from peptide design to validated monoclonal antibody.
Explore Hybridoma Antibody DevelopmentPeptide Synthesis | Polyclonal Antibody | Rabbit Single B Cell
Contact Us
+86-27-6552-3339
info@atagenix.com
Building C, R & D Building, No. 666, Shendun 4th Road, Donghu New Technology Development Zone, Wuhan

