AtaGenix Laboratories
AtaGenix Laboratories
Release time: 2025-10-14 View volume: 438
Project Snapshot — A plant biology team at Zhejiang University studying G protein-mediated crop growth regulation needed a phospho-specific antibody targeting PIP1;2 at Thr172 in tomato. AtaGenix delivered a custom anti-PIP1;2(pT172) polyclonal antibody with site-level discrimination (Thr172 vs. neighboring Tyr171), enabling discovery of the GGC1–CPK28–PIP1;2 signaling axis — published in Cell Reports.
Plant vigor directly determines crop yield and photosynthetic efficiency, representing a core focus of global food security research. The G protein signaling pathway is a key regulator of plant growth, but its molecular mechanisms remain incompletely understood. A study published in Cell Reports from Zhejiang University revealed that the G protein γ subunit GGC1, which underwent positive selection during tomato domestication, acts as a negative regulator of plant growth and photosynthesis by suppressing CPK28-mediated phosphorylation of aquaporin PIP1;2 at Thr172. This identified a previously uncharacterized signaling cascade — the GGC1–CPK28–PIP1;2 axis — with direct implications for high-yield crop breeding.
To verify the regulatory mechanism by which GGC1 influences CPK28-mediated PIP1;2 phosphorylation, the research team required an antibody that could: (1) specifically recognize PIP1;2 phosphorylated at Thr172 without cross-reacting with the unmodified form; (2) distinguish Thr172 phosphorylation from neighboring Tyr171 phosphorylation; (3) work in tomato (Solanum lycopersicum) tissue samples via Western Blot for quantitative detection; and (4) detect low-abundance phosphorylated protein with high sensitivity.
This project presented unique challenges beyond standard phospho-antibody development:
AtaGenix designed a targeted phospho-antibody workflow:
The custom anti-PIP1;2(pT172) antibody provided the critical experimental evidence enabling the research team to demonstrate that GGC1 negatively regulates photosynthetic efficiency and plant vigor by suppressing CPK28-mediated phosphorylation of PIP1;2 at Thr172. This discovery revealed a previously unknown G protein–mediated signaling cascade governing crop growth, with direct implications for breeding strategies to improve tomato yield through manipulation of the GGC1–CPK28–PIP1;2 pathway.
Figure 1. Functional divergence of G protein γ subunits in tomato. GGC1 suppresses CPK28-mediated phosphorylation of PIP1;2 at Thr172, negatively regulating plant vigor and photosynthesis.
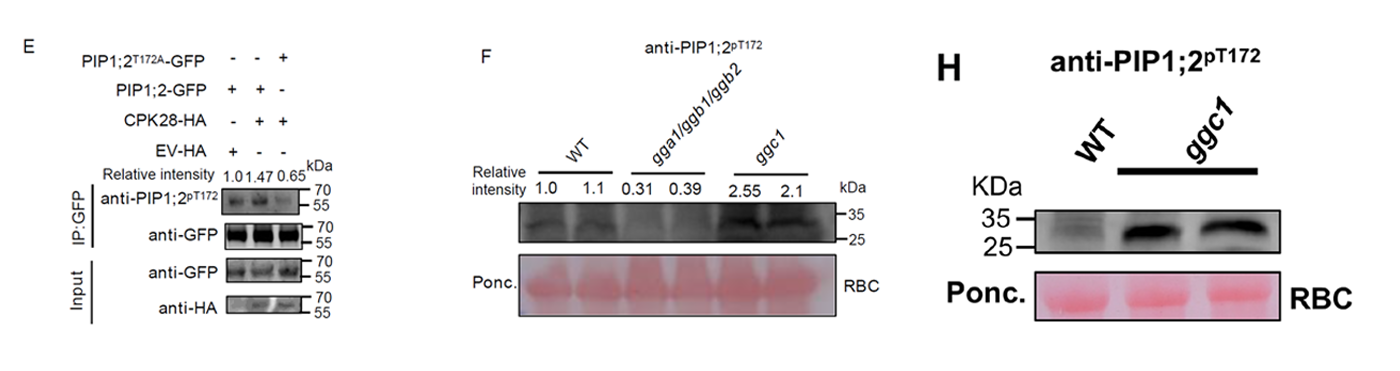
Figure 2. Western Blot validation of PIP1;2 Thr172 phosphorylation using AtaGenix custom phospho-specific antibody. Quantitative detection in tomato GGC1 mutant vs. wild-type samples confirmed the regulatory relationship.
About AtaGenix
AtaGenix provides one-stop customized antibody and protein solutions, from phospho-peptide design and two-round affinity purification through multi-platform validation. Our phospho-specific antibody service supports targets across mammalian, plant, and other species — not limited to commercially available targets. Learn more at www.atagenix.com.
This case study is based on a published research collaboration. Results may vary depending on target species, modification site, and experimental conditions. All proprietary client information is subject to NDA. Reference: Cell Reports. DOI: see original publication.
Need a phospho-specific antibody for a non-standard target — plant, insect, or unconventional species? AtaGenix designs custom phospho-peptides and delivers two-round purified antibodies validated for your specific application.
Explore Anti-PTM Antibody ServiceContact Us
+86-27-6552-3339
info@atagenix.com
Building C, R & D Building, No. 666, Shendun 4th Road, Donghu New Technology Development Zone, Wuhan

