AtaGenix Laboratories
AtaGenix Laboratories
Release time: 2025-12-24 View volume: 314
Service Overview — AtaGenix’s 3H E. coli expression platform (High Throughput, High Efficiency, High Quality) achieves >95% expression success rate across tens of thousands of delivered projects. Featuring optimized vectors (pET, pGEX series), specialized host strains (T7E, C41, Arctic, proprietary AtaGenix strains), and a complete expression–purification–endotoxin control pipeline, the system delivers purity ≥85% and endotoxin <0.1 EU/mL from milligram to gram scale — ISO 9001 & ISO 13485 certified.
>95% Success Rate
Optimized pET/pGEX vectors combined with T7E (high expression), C41 (toxic protein tolerance), Arctic (low-temp solubility), and proprietary AtaGenix strains tackle difficult-to-express proteins.
High-Throughput & Scalable
15 medium/large-scale shakers, 12 ultrasonic disruptors, and pilot-scale high-pressure homogenizers. Milligram research-grade to gram-scale pre-industrial production.
Publication-Grade QC
Purity ≥85%, endotoxin <0.1 EU/mL. Validated for WB, protein interaction (SPR/GST pull-down), cell-based assays, and in vivo animal studies.
End-to-End Custom Service
Gene optimization → vector construction → strain screening → induction optimization → purification → QC. Dedicated project manager with 1:1 technical support.
Below are three peer-reviewed publications where AtaGenix’s E. coli platform provided critical reagents — spanning plant science, entomology, and microbiology.
Background: This study cloned the key gene SDR3.1 (encoding the MODD protein) and elucidated the molecular mechanisms of pre-harvest sprouting (PHS) in rice, providing tools for genetic improvement of PHS resistance.
AtaGenix’s Role: Produced recombinant SDR3.1 protein via E. coli expression, then used it as an immunogen to develop a rabbit monoclonal anti-SDR3.1 antibody. The antibody performed at 1:10,000 dilution in WB, accurately detecting SDR3.1 expression and phosphorylation across rice varieties (ZH11, MJLXL) and transgenic plants — directly validating that SDR3.1 negatively regulates seed dormancy by inhibiting ABI5 transcriptional activity.
Published in Nature Communications (IF: 14.7, Q1) — DOI: 10.1038/s41467-024-45402-z
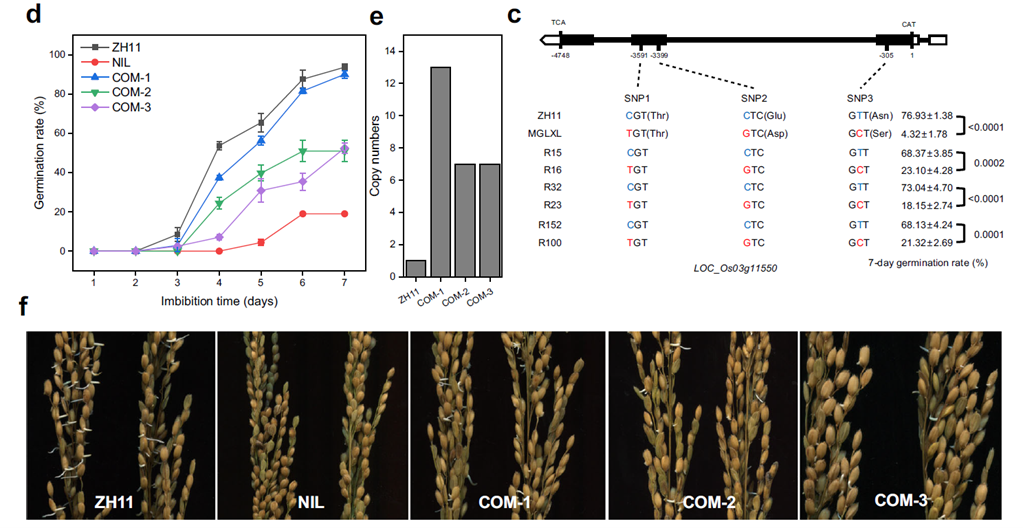
Fig. 1. Fine mapping of SDR3.1. Anti-SDR3.1 antibody produced from E. coli-expressed recombinant protein. Adapted from Nat Commun 2024.
Background: This research revealed how the transcription factor FTZ-F1 mediates non-fitness-cost resistance to Bt Cry1Ac toxin in Plutella xylostella, identifying new targets for eco-friendly pest control.
AtaGenix’s Role: Expressed recombinant FTZ-F1 protein in E. coli, then produced a rabbit polyclonal anti-FTZ-F1 antibody via Protein A/G purification. The antibody distinguished phosphorylated vs. non-phosphorylated FTZ-F1 by Phos-tag SDS-PAGE and performed at 1:10,000 in WB — confirming that MAPK-mediated FTZ-F1 phosphorylation drives differential Bt receptor gene expression.
Published in Nature Communications (IF: 14.7) — DOI: 10.1038/s41467-022-33706-x
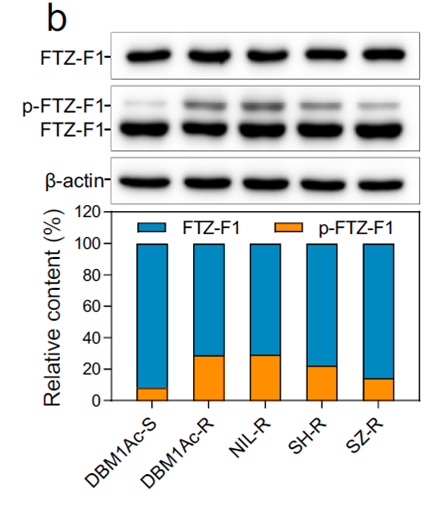
Fig. 2. FTZ-F1 protein expression and phosphorylation in larval midgut of five P. xylostella strains. Anti-FTZ-F1 antibody from E. coli-expressed antigen. Adapted from Nat Commun 2022.
Background: This study established a hypermutation system in Synechococcus elongatus PCC 7942, screened mutants tolerant to high-light and high-temperature (HLHT) stress, and dissected photosynthetic adaptation mechanisms under extreme conditions.
AtaGenix’s Role: Expressed and purified key proteins — RecA, UmuDC (DNA replication fidelity), and shikimate kinase (downstream validation) — via the E. coli system. These high-activity recombinant proteins supported hypermutation system construction and confirmed that shikimate kinase overexpression enhances HLHT tolerance by remodeling the photosynthetic chain and metabolic network.
Published in Nature Communications (IF: 14.7) — DOI: 10.1038/s41467-023-36964-5
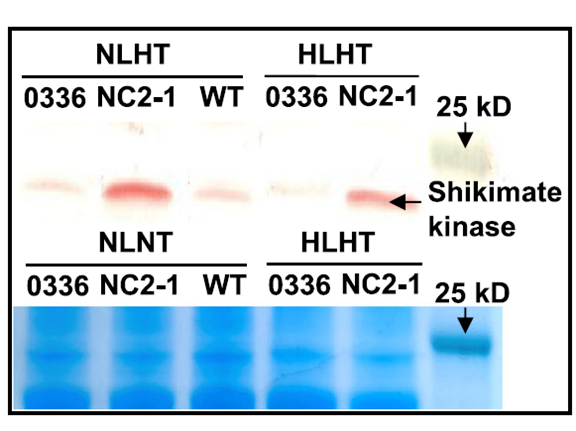
Fig. 3. WB (top) and SDS-PAGE (bottom) of shikimate kinase in HS199 strain. E. coli-expressed by AtaGenix. Adapted from Nat Commun 2023.
Proven at Scale
Tens of thousands of E. coli projects delivered over 15 years. Reagents cited in three Nature Communications papers across plant science, entomology, and microbiology.
Infrastructure Depth
15 shakers, 12 ultrasonic disruptors, pilot-scale homogenizers, and FPLC purification lines in a 3,000 m² facility. Standardized processes ensure batch-to-batch consistency.
Research-to-Industry
Flexible scale from mg-level custom proteins and antibody immunogens to gram-scale pilot production. Protocol adjustments based on protein solubility, toxicity, and modification needs.
Results may vary depending on target protein, construct design, and project scope. All proprietary client information is subject to NDA.
From recombinant protein production and antibody immunogen preparation to functional validation — AtaGenix’s 3H E. coli platform delivers reliable support at every stage.
Talk to Technical SupportResponse within 24 hours
Contact Us
+86-27-6552-3339
info@atagenix.com
Building C, R & D Building, No. 666, Shendun 4th Road, Donghu New Technology Development Zone, Wuhan

