AtaGenix Laboratories
AtaGenix Laboratories
Release time: 2025-12-24 View volume: 350
Service Overview — AtaGenix’s baculovirus–insect cell expression system (BEVS) delivers properly folded, post-translationally modified recombinant proteins for research and preclinical applications. Built on optimized Bac-to-Bac technology with Sf9, Sf21, and Hi5 host lines, the platform supports milligram-to-gram scale production with short turnaround, high throughput, and full-process QC — backed by 15 years of project delivery experience (ISO 9001 & ISO 13485 certified).
Eukaryotic Protein Expression
Proper folding and post-translational modifications (glycosylation, phosphorylation) for complex proteins — enzymes, antibodies, transcription factors — with biological activity consistent with native forms.
Flexible Vector & Host Options
Multiple baculovirus vectors paired with Sf9, Sf21, or Hi5 cell lines. Supports large gene inserts and full-length gene expression with optimized codon strategies.
Scalable Production
From milligram research-grade to gram-scale pre-industrial quantities. No CO₂ incubator required; strong contamination resistance reduces cost and timeline.
End-to-End Service
Gene optimization → vector construction → virus packaging → expression & purification → QC (purity, activity, endotoxin). Dedicated project manager throughout.
Below are four peer-reviewed publications where AtaGenix’s BEVS platform provided critical reagents — spanning oncology, tumor immunology, neuroscience, and gynecological oncology.
Background: This study clarified how DNA polymerase POLD1 drives bladder cancer proliferation and metastasis through a positive feedback loop with MYC, revealing a new diagnostic and therapeutic target.
AtaGenix’s Role: Produced high-activity GST-MYC recombinant protein via BEVS. Used as the core reagent in GST pull-down assays, it directly validated the specific binding of POLD1 to the MYC homology box 1 domain, confirming that POLD1 stabilizes MYC by competitively binding FBXW7.
Published in Nature Communications (IF: 14.7) — DOI: 10.1038/s41467-023-38160-x
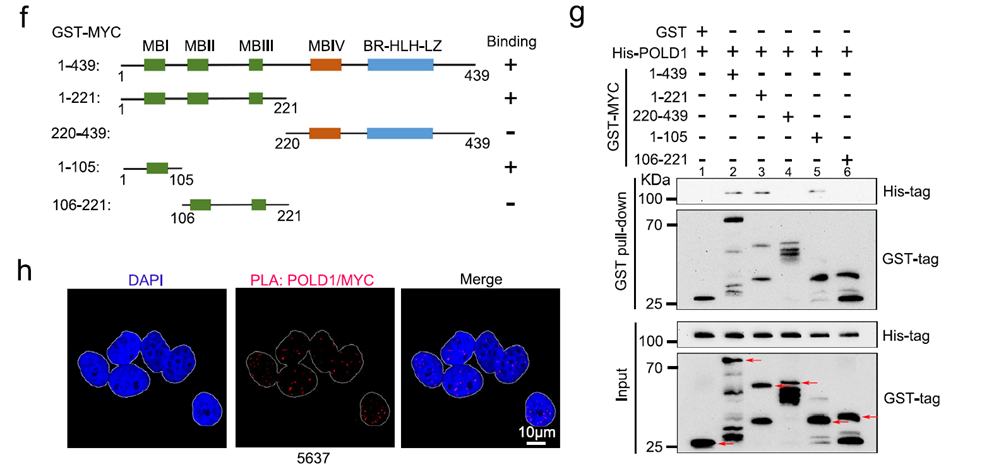
Fig. 1. POLD1 is a direct binding partner of MYC. GST pull-down assay using AtaGenix-produced GST-MYC protein. Adapted from Wen et al., Nat Commun 2023.
Background: Targeting high PADI4 expression in breast cancer, this study developed monoclonal antibodies to inhibit fibronectin citrullination and improve the tumor microenvironment.
AtaGenix’s Role: Constructed and screened high-specificity anti-human PADI4 monoclonal antibodies via hybridoma technology. The antibodies inhibited fibronectin citrullination, reduced EMT-related gene expression, and significantly suppressed breast cancer cell proliferation and migration in both in vitro and in vivo models.
Published in Biomedicine & Pharmacotherapy (IF: 6.9) — DOI: 10.1016/j.biopha.2022.113289
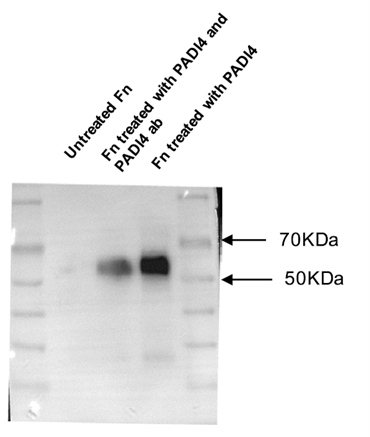
Fig. 2. Identification of citrullination of fibronectin using AtaGenix-produced anti-PADI4 mAb. Adapted from Zhu et al., Biomed Pharmacother 2022.
Background: This study investigated the SERBP1-PCIF1 complex in m⁶Am modification and its regulatory role in neuropathic pain with comorbid anxiety, identifying new therapeutic targets.
AtaGenix’s Role: Expressed and purified the recombinant PCIF1-MTase domain protein in vitro. The high-purity protein was used in methyltransferase activity assays, validating the synergistic catalytic effect of SERBP1-PCIF1 interaction on m⁶Am modification and providing key evidence for the “SERBP1-PCIF1-Maf1” regulatory axis.
Published in Nature Communications (IF: 14.7) — DOI: 10.1038/s41467-025-62565-5
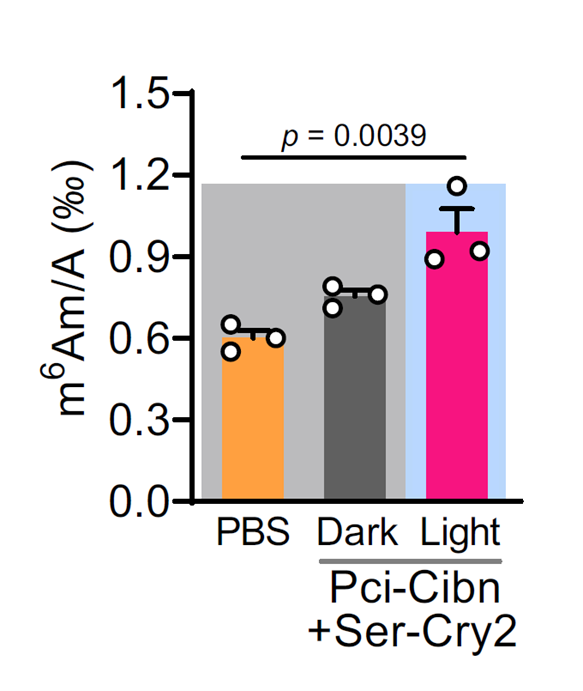
Fig. 3. SERBP1 and PCIF1 interact and co-catalyze m⁶Am modifications on mRNA. Adapted from Nat Commun 2025.
Background: This study targeted the glycolytic enzyme ENO1 in cervical cancer, developing monoclonal antibodies to block ENO1-mediated plasminogen activation and inhibit tumor invasion.
AtaGenix’s Role: Expressed and purified ENO1 protein using the Sf9 insect cell baculovirus system for mouse immunization, then constructed and screened five high-activity hybridoma cell lines. The resulting ENO1 mAbs blocked plasminogen activation, inhibiting cervical cancer cell migration and colony formation — establishing the foundation for nanocarrier-mediated targeted therapy.
Published in American Journal of Cancer Research — Source: ajcr.us/article/view/129535
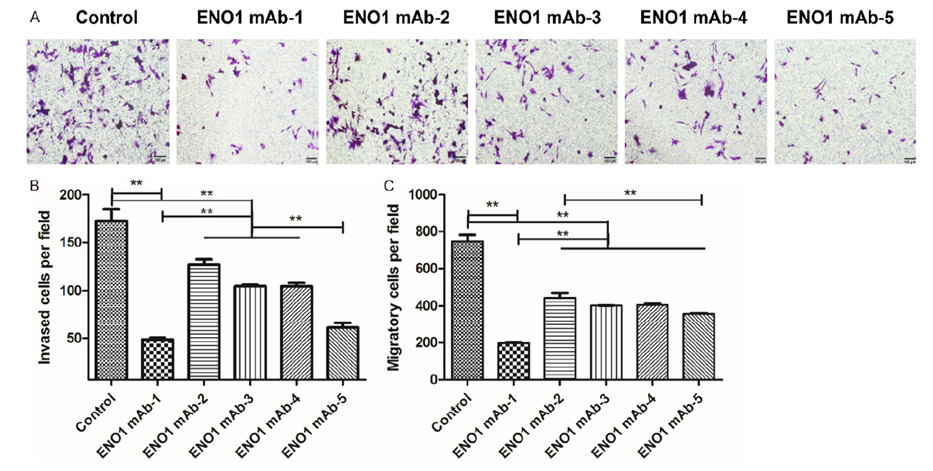
Fig. 4. ENO1 mAbs inhibit invasion and migration of cervical cancer cells. Sf9/BEVS-expressed ENO1 protein was used for immunization and hybridoma screening. Adapted from Am J Cancer Res.
Proven Track Record
15 years of protein expression experience. BEVS-produced reagents cited in Nature Communications, Biomedicine & Pharmacotherapy, and other peer-reviewed journals.
Domain Expertise
Deep specialization in complex structural proteins and antibodies for oncology, neuroscience, immunology, and infectious disease applications.
Quality Certified
ISO 9001:2015 & ISO 13485:2016 certified. Every batch includes purity (SDS-PAGE/SEC), activity, and endotoxin documentation.
Flexible & Scalable
Customizable yield, purity, and timeline from basic research to preclinical stage. Expression strategy optimized per project with dedicated PM support.
Results may vary depending on target protein, construct design, and project scope. All proprietary client information is subject to NDA.
Whether you need recombinant protein production, monoclonal antibody development, or protein interaction validation via BEVS — AtaGenix delivers end-to-end support from gene to purified protein.
Talk to Technical SupportResponse within 24 hours
Contact Us
+86-27-6552-3339
info@atagenix.com
Building C, R & D Building, No. 666, Shendun 4th Road, Donghu New Technology Development Zone, Wuhan

