AtaGenix Laboratories
AtaGenix Laboratories
Release time: 2025-12-19 View volume: 344
Service Overview — AtaGenix’s mammalian cell expression platform has delivered 5,000+ projects and 200+ stable cell line constructions over 15 years. Offering HEK293 and CHO-S transient expression (mL to 100 L), stable cell line development (CHO-K1, DG44), and gram-scale recombinant antibody production, the system achieves native conformation, full glycosylation, and endotoxin <0.1 EU/mL in Class 100,000 cleanroom facilities — ISO 9001 & ISO 13485 certified.
5,000+ Projects Delivered
Over a decade of mammalian expression expertise. In-house high-yield systems increase protein yields 3–6× over standard protocols. 200+ stable cell lines constructed.
Transient & Stable Expression
HEK293 and CHO-S serum-free suspension culture (mL to 100 L transient). CHO-K1/DG44 stable cell line development. Gram-scale recombinant antibody production.
Cleanroom & Scalable
Class 100,000 clean cell culture facilities. Endotoxin <0.1 EU/mL. Serum-free suspension batches up to 200 L+, bridging lab-scale research and industrial production.
End-to-End Service
Gene optimization → structure analysis → expression strategy → transfection → purification → QC & data analysis. Dedicated project manager throughout.
Below are four peer-reviewed publications where AtaGenix’s mammalian expression platform provided critical reagents — spanning tumor immunology, vaccine development, and neuroscience.
Background: This study developed a trained immunity-related vaccine (TIrV) based on bacterial outer membrane vesicles (OMVs), targeting tumor-associated macrophages (TAMs) to overcome antigen dependence and immune tolerance limitations of traditional tumor vaccines.
AtaGenix’s Role: Expressed and purified SpyCatcher-RBD fusion protein via mammalian cells, enabling precise OMV surface modification with SIRPα-Fc via SpyTag/SpyCatcher ligation. The fully functional protein facilitated construction of OMV-SIRPα@CaP/GM-CSF nano-hybrids, driving myeloid progenitor training and TAM phagocytosis activation. Significant anti-tumor effects validated in MC38 and B16-F10 models.
Published in Advanced Materials (IF: 27.4) — DOI: 10.1002/adma.202306158
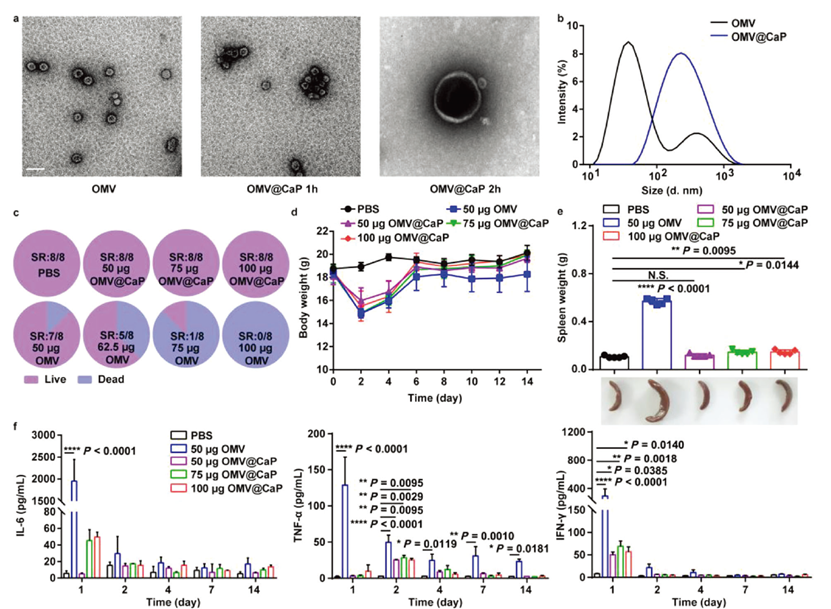
Fig. 1. OMV@CaP development and attenuated adverse effects. SpyCatcher-RBD fusion protein expressed by AtaGenix. Adapted from Adv Mater 2024.
Background: To overcome insufficient immunogenicity of RBD subunit vaccines, this study used icosahedral DNA origami (ICO) as a scaffold for virus-mimetic multimeric nanovaccines, optimizing B-cell activation efficiency.
AtaGenix’s Role: Expressed prototype RBD-SpyCatcher fusion and Omicron variant RBD (His-tagged) via mammalian cells, retaining native conformation with >85% loading efficiency onto DNA origami scaffolds. The nanovaccine (ICO-RBD) elicited potent humoral and T-cell immunity, with neutralizing titers significantly exceeding soluble RBD and cross-protection against multiple variants.
Published in Nature Communications (IF: 14.7) — DOI: 10.1038/s41467-024-53937-4
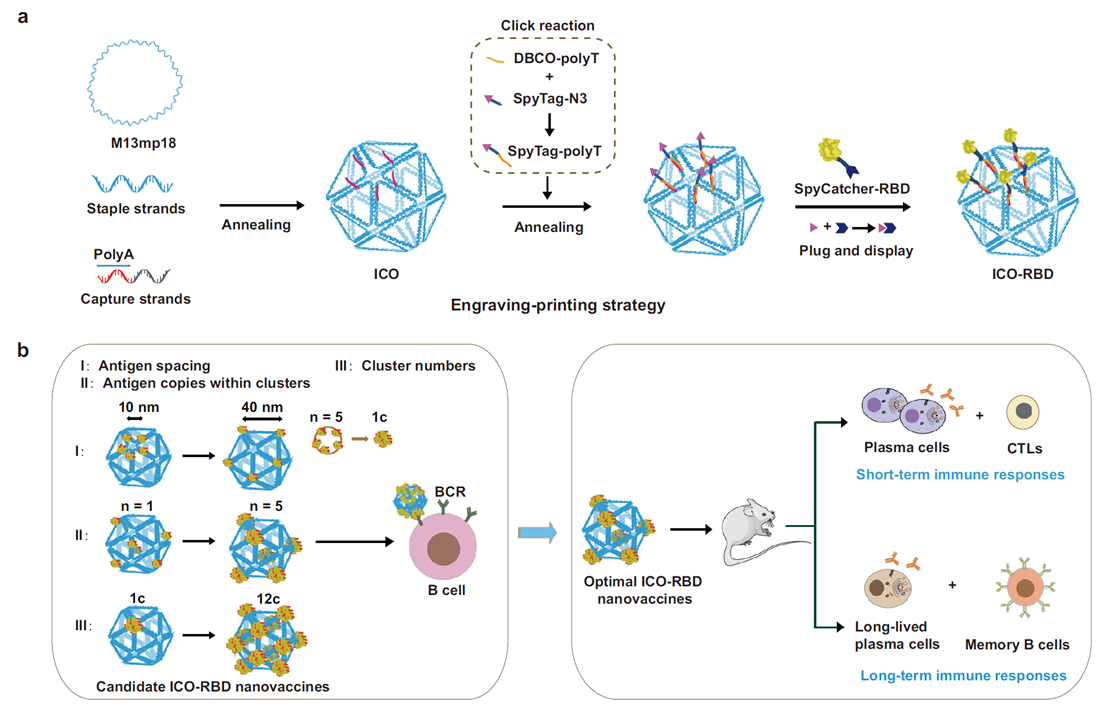
Fig. 2. Design and construction of ICO-RBD nanovaccines. RBD-SpyCatcher expressed by AtaGenix mammalian platform. Adapted from Nat Commun 2024.
Background: This study explored how meningeal autoreactive B cells drive neuroinflammatory relapse in the EAE mouse model, providing new therapeutic targets for multiple sclerosis (MS).
AtaGenix’s Role: Supplied recombinant mouse MOG protein with native-like conformation, enabling effective activation of autoreactive B–T cell cognate interactions in the meningeal microenvironment. The protein helped elucidate how meningeal B cells drive neuroinflammation via MHC II-mediated antigen presentation and GM-CSF-dependent neutrophil recruitment.
Published in Immunity (IF: 25.5) — DOI: 10.1016/j.immuni.2025.06.016
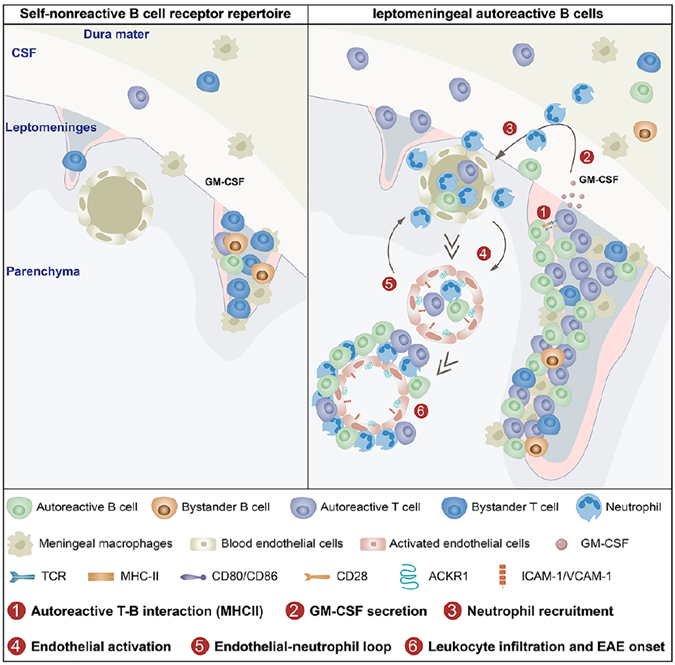
Fig. 3. Meningeal B cell interactions drive neuroinflammatory relapse. Recombinant mouse MOG protein provided by AtaGenix. Adapted from Immunity 2025.
Background: This work revealed the complement C3/C3aR pathway’s role in perioperative neurocognitive disorder (PND) in elderly patients, and explored platelet factor 4 (PF4) as a neuroprotective mechanism.
AtaGenix’s Role: Custom-expressed the C3-selective inhibitor CR2-crry protein via mammalian cells. The fully functional fusion protein precisely inhibited C3 activity, confirming that C3 blockade reduces glial activation, restores PF4 levels, and improves cognitive function — supporting ceftriaxone’s clinical potential for PND prevention.
Published in Molecular Psychiatry (IF: 9.6) — DOI: 10.1038/s41380-025-03103-z
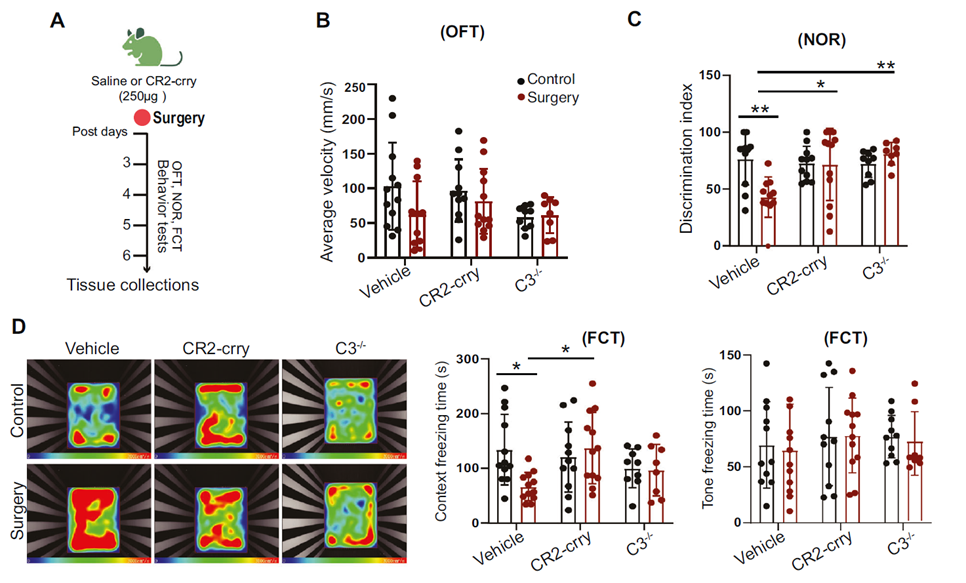
Fig. 4. C3 blockage alleviates cognitive decline in PND mouse model. CR2-crry protein expressed by AtaGenix. Adapted from Mol Psychiatry 2025.
Top-Journal Validated
Proteins cited in Advanced Materials (IF 27.4), Immunity (IF 25.5), Nature Communications (IF 14.7), and Molecular Psychiatry (IF 9.6).
Complex Protein Expertise
Specializing in fusion proteins (SpyCatcher-RBD), autoantigens (MOG), complement inhibitors (CR2-crry), and other challenging constructs requiring native conformation and full glycosylation.
Research to Industry
From mg-scale custom proteins for basic research to 200 L+ serum-free batch production for industrialization. Class 100,000 cleanroom, ISO certified.
Results may vary depending on target protein, construct design, and project scope. All proprietary client information is subject to NDA.
From recombinant protein production and antibody development to vaccine antigen preparation — AtaGenix’s mammalian expression platform delivers native-conformation, publication-grade proteins at any scale.
Talk to Technical SupportResponse within 24 hours
Contact Us
+86-27-6552-3339
info@atagenix.com
Building C, R & D Building, No. 666, Shendun 4th Road, Donghu New Technology Development Zone, Wuhan

