AtaGenix Laboratories
AtaGenix Laboratories
Release time: 2024-11-13 View volume: 1999
Project Snapshot — AtaGenix expressed ENO1 protein via the Sf9 insect cell baculovirus system for mouse immunization, then constructed and screened five high-activity hybridoma cell lines producing anti-ENO1 monoclonal antibodies (ENO1mAbs). The antibodies blocked plasminogen activation, inhibited glycolysis, and suppressed cervical cancer cell invasion, proliferation, and colony formation — establishing the foundation for subsequent folic acid-conjugated PLGA nanoparticle-mediated targeted therapy.
Published in American Journal of Cancer Research — Source: ajcr.us/article/view/129535
α-Enolase (ENO1) is a glycolytic enzyme that also functions on the cell surface as a plasminogen receptor, promoting extracellular matrix degradation and tumor cell invasion. ENO1 is highly expressed in cervical cancer and correlates with poor prognosis. This study aimed to develop monoclonal antibodies against ENO1 to block both its surface plasminogen-binding and intracellular glycolytic functions, then enhance intracellular delivery via folic acid-conjugated PLGA nanoparticles.
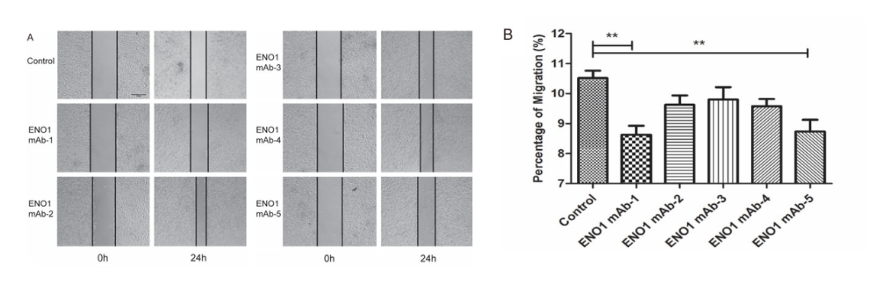
Fig. 1. Study overview: ENO1 monoclonal antibody development and functional validation in cervical cancer models. Adapted from Am J Cancer Res.
Protein Expression: ENO1 protein was expressed and purified using the Sf9 insect cell baculovirus system (BEVS), producing the immunogen for mouse immunization.
Hybridoma Screening: Five high-activity hybridoma cell lines were constructed and screened, yielding ENO1 monoclonal antibodies with validated specificity against human ENO1.
Functional Outcome: ENO1mAbs blocked surface plasminogen activation (inhibiting migration and invasion), suppressed intracellular glycolysis (reducing lactate and pyruvate), and inhibited proliferation and colony formation in SiHa and HeLa cervical cancer cell lines.
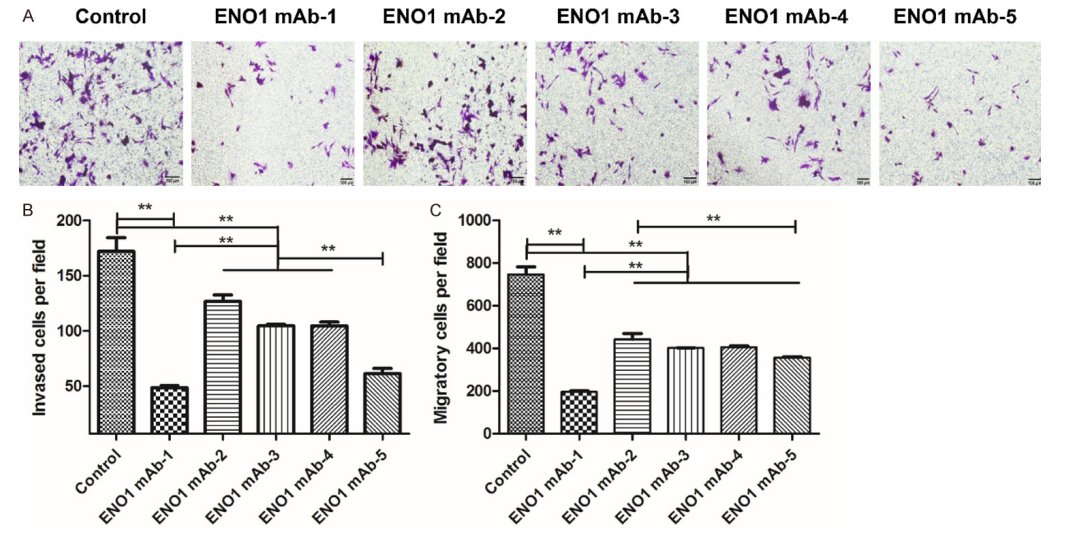
Fig. 2. ENO1mAb inhibitory effects on SiHa cells: reduced invasion, proliferation, and colony formation. Antibody produced from Sf9-expressed ENO1 immunogen by AtaGenix. Adapted from Am J Cancer Res.
5
Hybridoma Lines Screened
Sf9/BEVS
Expression System
ENO1
Glycolytic Target
SiHa + HeLa
Cell Lines Validated
Why This Matters
This study demonstrates a dual-mechanism approach to cervical cancer therapy: ENO1mAbs simultaneously block extracellular plasminogen activation (inhibiting invasion) and intracellular glycolysis (starving tumor metabolism). The subsequent integration with FA-SS-PLGA nanoparticles for intracellular delivery represents a promising platform for targeted antibody therapy. AtaGenix’s end-to-end contribution — from Sf9 protein expression to hybridoma screening of five functional clones — provided the essential antibody reagents that made this discovery possible.
Results may vary depending on target, expression system, and project scope. All proprietary client information is subject to NDA.
Need custom monoclonal antibodies from antigen expression to hybridoma screening? AtaGenix provides end-to-end development with BEVS, mammalian, and E. coli expression platforms.
Talk to Technical SupportResponse within 24 hours
Contact Us
+86-27-6552-3339
info@atagenix.com
Building C, R & D Building, No. 666, Shendun 4th Road, Donghu New Technology Development Zone, Wuhan

